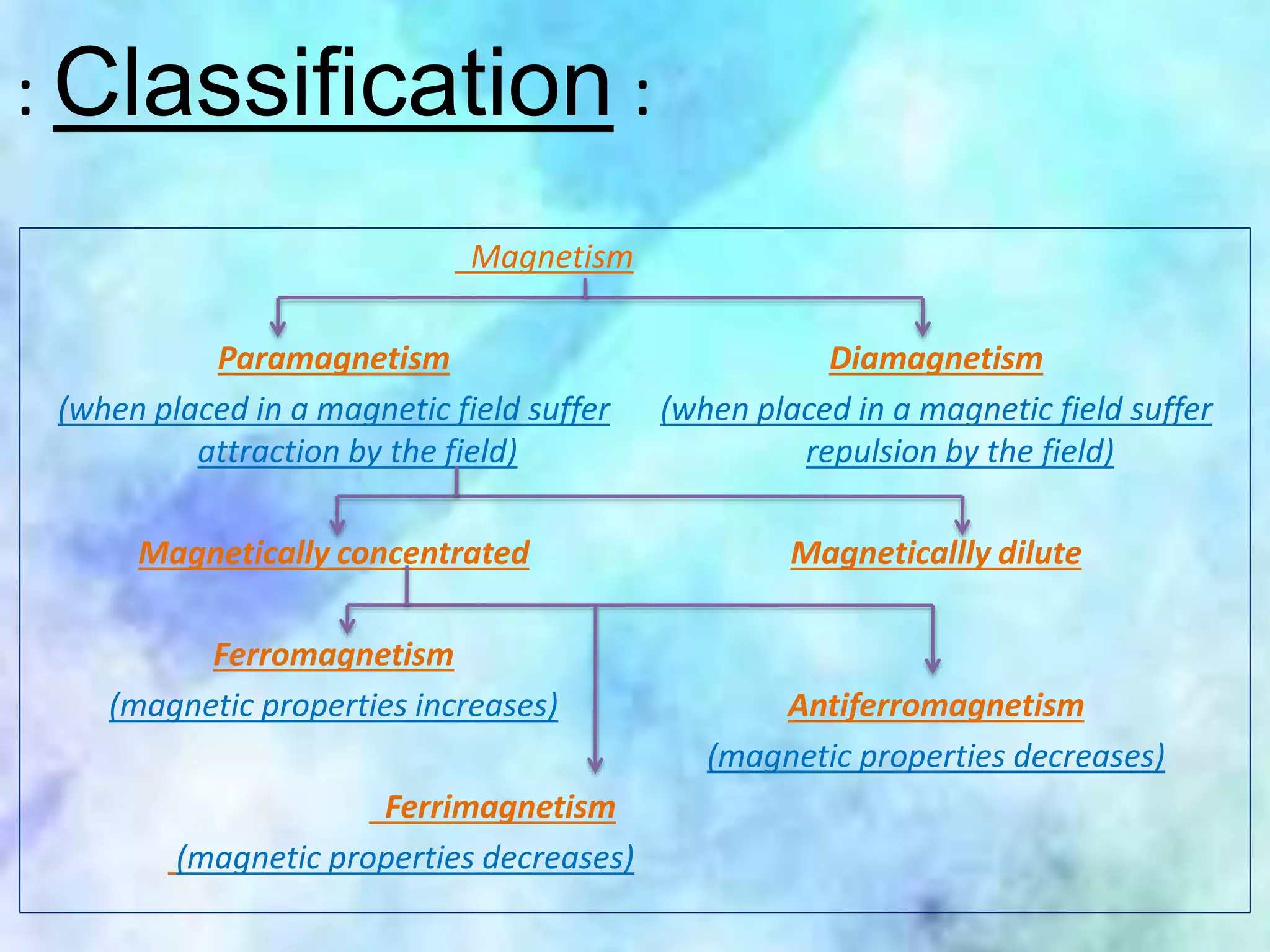
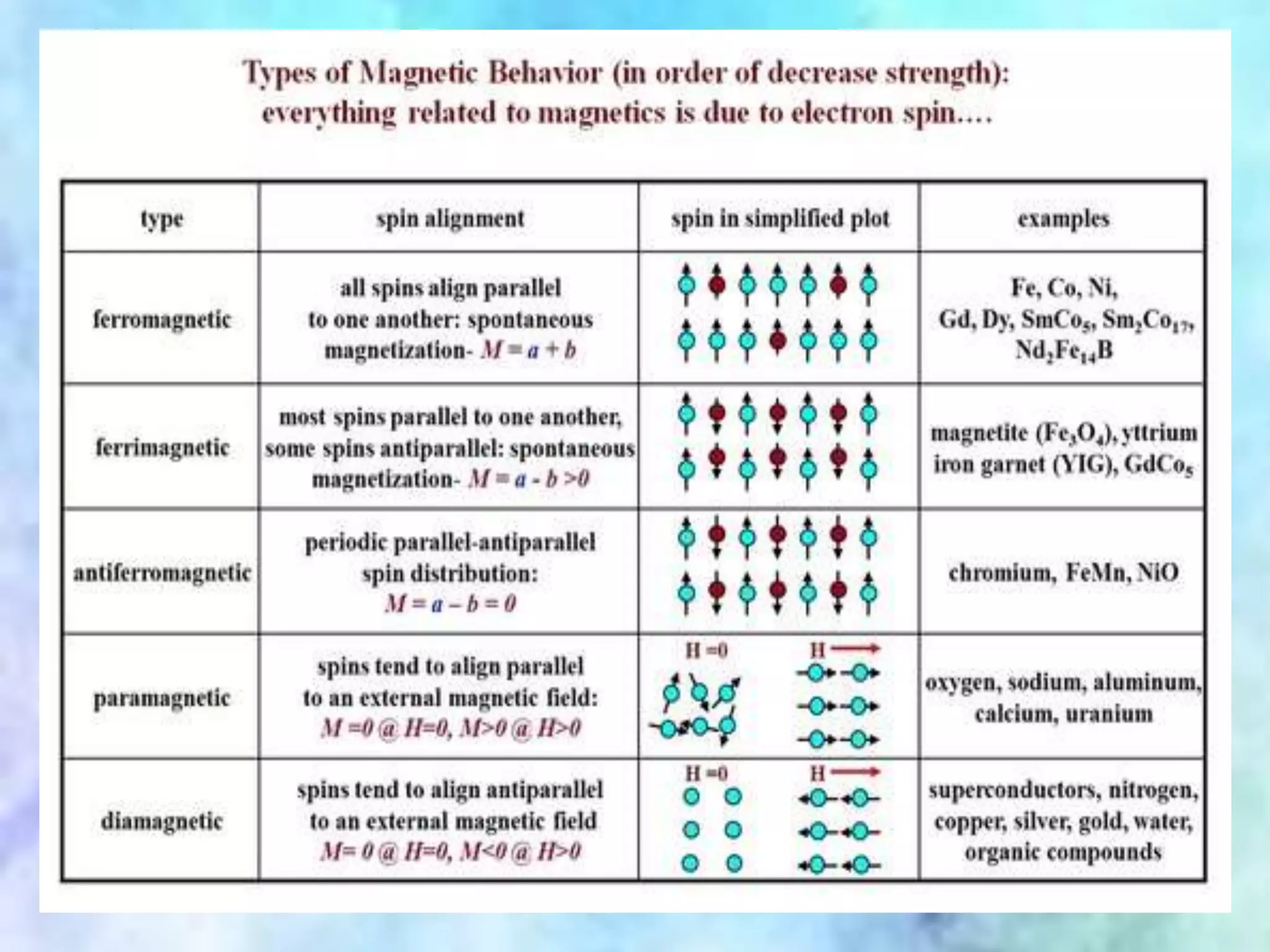
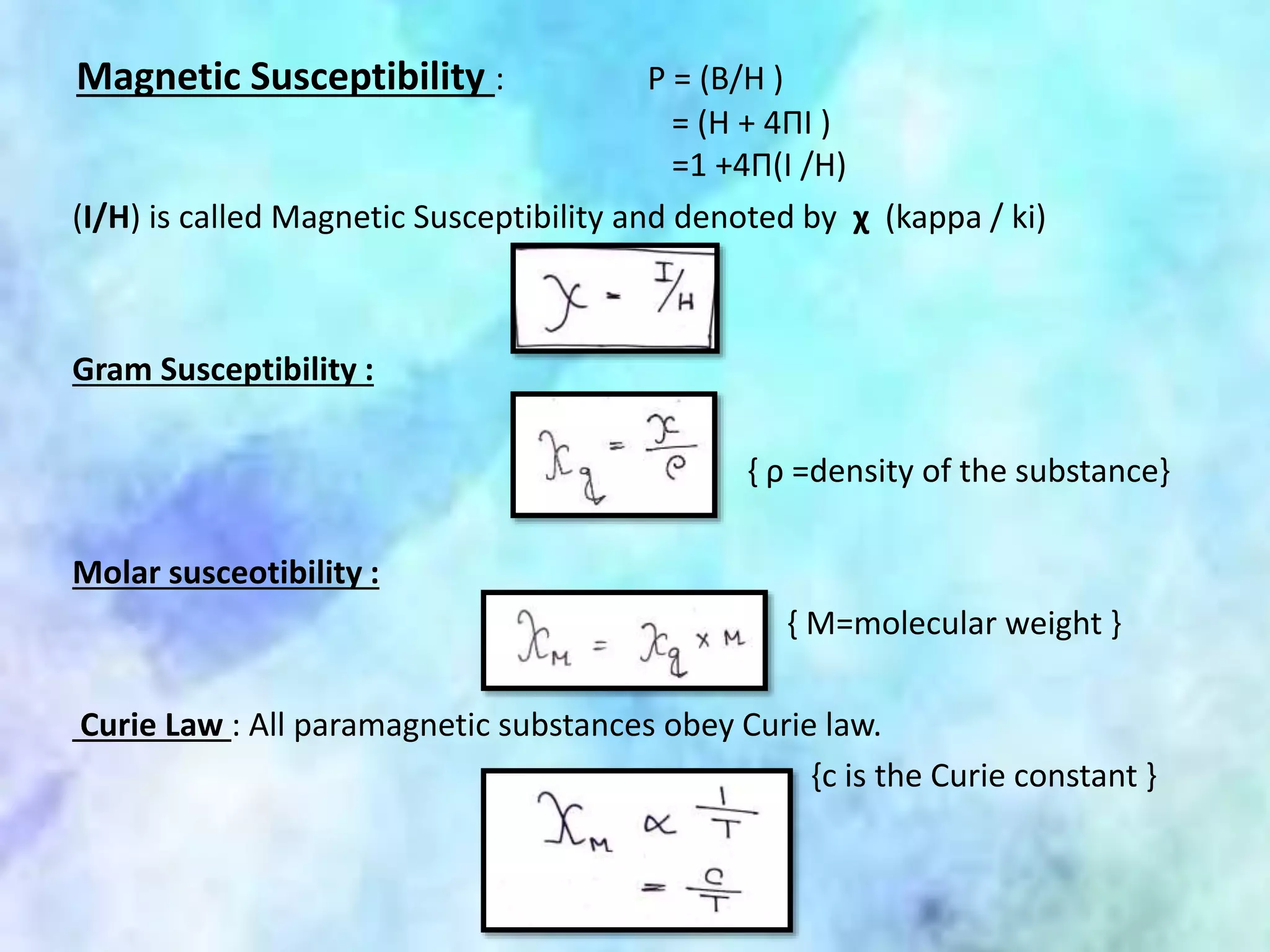
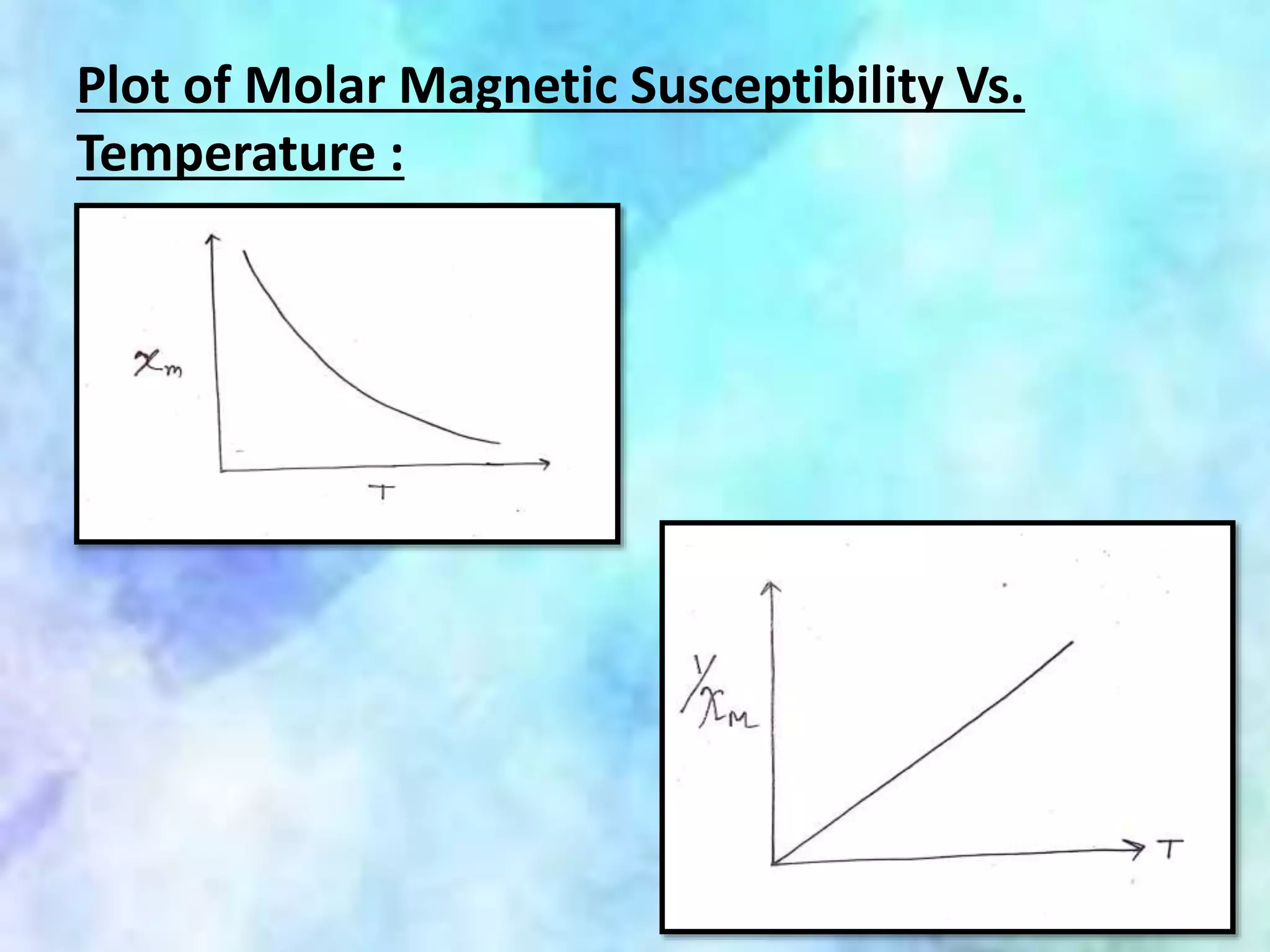
This document summarizes magnetic properties of transition metal complexes. It defines different types of magnetism including paramagnetism, ferromagnetism, ferrimagnetism, and diamagnetism. It describes how orbital and spin motions of electrons give rise to magnetic moments. It provides equations to calculate theoretical magnetic moments based on spin and orbital contributions. It also discusses magnetic susceptibility, Curie's law relating susceptibility and temperature, and Gouy's method for measuring susceptibility by observing a sample's movement in a magnetic field.