Differential WBC Count
- 1. Dr. S. MEENATCHISUNDARAM ASSOCIATE PROFESSOR DEPARTMENT OF MICROBIOLOGY SNMV COLLEGE OF ARTS AND SCIENCE COIMBATORE https://orcid.org/0000-0002-8691-449X 95496 https://scholar.google.com/citations?user=IkdZ5XsAAAAJ&hl=en DIFFERENTIAL WHITE CELL COUNT
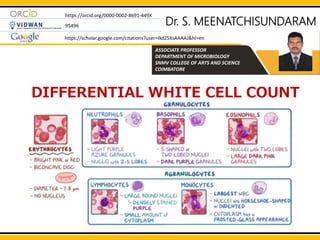
- 2. INTRODUCTION A differential white cell count provides information on the different white cells present in the circulating blood, i.e. neutrophils, lymphocytes, monocytes, eosinophils, basophils (rarely seen). Providing the total WBC count is known, the absolute number of each white cell type, i.e. number of each cell per litre of blood, can be calculated and an assessment made of whether the number of a particular cell type is increased or decreased DIFFERENTIAL WHITE CELL COUNT
- 3. Blood must be collected with care and adequate safety precautions to ensure test results are reliable, contamination of the sample is avoided and infection from blood transmissible pathogens is prevented. Protective gloves should be worn when collecting and handling blood samples. Lancets, needles, and syringes must be sterile, and dry, and blood collecting materials must be discarded safely to avoid injury from needles and lancets. CAPILLARY BLOOD Capillary blood is mainly used when the patient is an infant or young child and the volume of blood required is small, e.g. to measure haemoglobin, perform a WBC count, and to make thick and thin blood films. Thick blood films for malaria parasites are best made from capillary blood (anticoagulated blood is more easily washed from slides during staining). Capillary blood can be collected from The ‘ring’ finger of a child or adult The heel of an infant up to one year old DIFFERENTIAL WHITE CELL COUNT
- 4. TECHNIQUE FOR COLLECTING CAPILLARY BLOOD Cleanse the puncture area with 70% ethanol. Allow the area to dry. Using a sterile pricker or lancet, make a rapid puncture, sufficiently deep to allow the free flow of blood. Wipe away the first drop of blood with a dry piece of cotton wool and use the next few drops for the test. Do not squeeze too hard because this will result in an unreliable test result. When sufficient blood has been collected, press a piece of dry cotton wool over the puncture area until bleeding stops. COLLECTION OF CAPILLARY BLOOD FROM A FINGER COLLECTION OF BLOOD FROM THE HEEL OF AN INFANT DIFFERENTIAL WHITE CELL COUNT
- 5. Place a drop of blood on the end of a clean dry slide. Avoid making the drop too large (if too large, use a drop from the excess blood to make the film) Using a clean smooth edged spreader, draw the spreader back to touch the drop of blood and allow the blood to extend along the edge of the spreader. Holding the spreader at an angle of about 30, spread the drop of blood to make a film about 40–50 mm in length (two thirds of the slide) DIFFERENTIAL WHITE CELL COUNT
- 6. Wipe clean the end of the spreader. Immediately air dry the film by waving the slide back and forth. Protect the dried film from dust and insects When completely dry and within a few minutes of making the blood film, fix it in absolute methanol Features of a well made film ● Not too thick, nor too long ● Free from lines and holes ● Has a smooth ‘tail’ DIFFERENTIAL WHITE CELL COUNT
- 7. METHANOL FIXING THIN BLOOD FILMS When completely dry, fix a blood film with absolute methanol (methyl alcohol). Place the film on a staining rack and add 1–2 drops of moisture-free methyl alcohol and allow it to dry on the film. Alternatively, the blood film can be immersed in a container of absolute methanol for about 2 minutes, but this is a more expensive method of fixing and also in tropical humid climates there is a greater risk of the alcohol absorbing water from the atmosphere which will result in poor fixing of blood cells. Water-free methanol Satisfactory fixing of thin blood films requires the use of water-free absolute methanol. Poorly fixed cells due to methanol containing water When absolute methanol is not available, absolute ethanol (ethyl alcohol) can be used but this is more expensive and usually less available than methanol DIFFERENTIAL WHITE CELL COUNT
- 8. LEISHMAN STAINING TECHNIQUE Method Cover the blood film (preferably methanol prefixed) with undiluted stain but do not flood the slide. If using a dropper bottle count the number of drops required to cover the film. Add twice the volume of pH 6.8 buffered water (i.e. twice the number of drops as stain). The diluted stain should not overflow. Ensure the water is well mixed with the stain by blowing on the diluted stain or mixing the stain and water using a plastic bulb pipette. Allow to stain for 10 minutes Wash off the stain with tap water* (filtered if not clean). Do not tip off the stain, because this will leave a fine deposit covering the film. Wipe the back of the slide clean and stand it in a draining rack for the smear to dry. The blood film should appear neither too pink nor too blue Tap water: If the tap water is highly acidic, resulting in too pink a blood film or highly alkaline, resulting in too blue a blood film, try using boiled cooled water or filtered rain water. If neither of these are suitable, wash the film with pH 6.8 buffered water. DIFFERENTIAL WHITE CELL COUNT
- 9. STAINING RESULTS Red cells . . . . . . . . . . . . . . . . . . . . . . . . . . . . Pink-red Nucleus of cells . . . . . . . . . . . . . . . . . . . Purple-violet Cytoplasm Neutrophils, eosinophils . . . . . . . . . . . . . . Pale pink Large lymphocytes . . . . . . . . . . . . . . . . . . Clear blue Small lymphocytes . . . . . . . . . . . . Darker clear blue Monocytes . . . . . . . . . . . . . . . . . . . . . . . . . Grey-blue Granules Eosinophils . . . . . . . . . . . . . . . . . . . . . . . Orange-red Neutrophils . . . . . . . . . . . . . . . . . . . . . Mauve-purple Toxic granules. . . . . . . . . . . . . . . . . . . . . . Dark violet Basophils. . . . . . . . . . . . . . . . . . . . . . Dark blue-violet Platelets . . . . . . . . . . . . . . . . . . . . . . . . . . Purple-blue Inclusions Malaria pigment (in monocytes) . . . . . . Brown-black Howell-Jolly body. . . . . . . . . . . . . . . . . . Purple-violet Auer body (in myeloblast) . . . . . . . . . . . . Purple-red DIFFERENTIAL WHITE CELL COUNT
- 10. Thin blood film is prepared and stained. Allow the stained film to dry completely before examining it. Keep the prepared glass slide under low power objective lens Then identify different types of WBC under medium power Draw a table with 10 boxes both on horizontal and vertical axis on the observation book Fix the slide on the platform and choose a area towards the corner Note the different types of WBC found on the table in an abbreviated Move downward and in a chin like manner till 100 cells are observed After counting 100 cells prepare the report DIFFERENTIAL WHITE CELL COUNT
- 13. REPORT Neutrophils - 61% Lymphocytes - 29% Eosinophils - 5% Monocytes - 5% Basophils - 0 TOTAL - 100 DIFFERENTIAL WHITE CELL COUNT
- 14. INTRODUCTION Calculate the absolute number of each white cell type by multiplying the number of each cell counted (expressed as a decimal fraction) by the total WBC count. Example If: Percentage of neutrophils counted = 80% i.e. 0.80 when expressed as a decimal fraction. If total WBC count = 8.6 x 109/L Absolute neutrophil count is 0.80 x 8.6 = 6.9x 109/L DIFFERENTIAL WHITE CELL COUNT
Editor's Notes
- This template was inserted from Power-user, the productivity add-in for PowerPoint and Excel. Get thousands of templates, icons, maps, diagrams and charts with Power-user. Visit www.powerusersoftwares.com!
