Information-Cards.pptx
•Download as PPTX, PDF•
0 likes•6 views
Giant covalent structure
Report
Share
Report
Share
Recommended
More Related Content
Similar to Information-Cards.pptx
Similar to Information-Cards.pptx (20)
Introduction to Nano science and Nanotechnology Part 4

Introduction to Nano science and Nanotechnology Part 4
More from JinsyAjish2
More from JinsyAjish2 (9)
C1.23 - Crude Oil and start of Distillation column.ppt

C1.23 - Crude Oil and start of Distillation column.ppt
6.1-a)---b)-Redox-Processes--Half-Equations-and-Oxidation-States.pptx

6.1-a)---b)-Redox-Processes--Half-Equations-and-Oxidation-States.pptx
Recently uploaded
9953330565 Low Rate Call Girls In Rohini Delhi NCR

9953330565 Low Rate Call Girls In Rohini Delhi NCR9953056974 Low Rate Call Girls In Saket, Delhi NCR
Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝9953056974 Low Rate Call Girls In Saket, Delhi NCR
Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝9953056974 Low Rate Call Girls In Saket, Delhi NCR
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
9953330565 Low Rate Call Girls In Rohini Delhi NCR

9953330565 Low Rate Call Girls In Rohini Delhi NCR
Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝
Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝
Capitol Tech U Doctoral Presentation - April 2024.pptx

Capitol Tech U Doctoral Presentation - April 2024.pptx
Procuring digital preservation CAN be quick and painless with our new dynamic...

Procuring digital preservation CAN be quick and painless with our new dynamic...
Blooming Together_ Growing a Community Garden Worksheet.docx

Blooming Together_ Growing a Community Garden Worksheet.docx
Difference Between Search & Browse Methods in Odoo 17

Difference Between Search & Browse Methods in Odoo 17
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️
Information-Cards.pptx
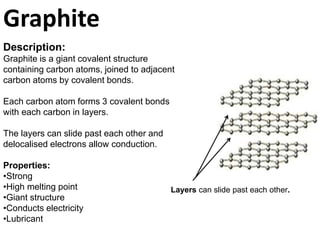
- 1. Graphite Description: Graphite is a giant covalent structure containing carbon atoms, joined to adjacent carbon atoms by covalent bonds. Each carbon atom forms 3 covalent bonds with each carbon in layers. The layers can slide past each other and delocalised electrons allow conduction. Properties: •Strong •High melting point •Giant structure •Conducts electricity •Lubricant Layers can slide past each other.
- 2. Diamond Description: Diamond is a giant covalent structure containing carbon atoms, joined to adjacent carbon atoms by covalent bonds. Each carbon atom forms 4 covalent bonds with each carbon. Making it hard but non-conductive. Properties: •Strong •High melting point •Giant structure •Lattice Structure •Non-conductive Each carbon forms 4 covalent bonds
- 3. Buckminster Fullerenes Description: Carbon-60 or the Buckminster Fullerene is a closed carbon cage that was isolated and synthesised from common soot. Properties: •Strong (stronger than diamond) •Stable (maintain shape) •High melting point •High boiling point •1 nanometre in diameter (1nm) ‘Bucky Ball’
- 4. Carbon Nanotubes Description: Carbon Nanotubes or CNT’s are flexible carbon tubes, synthesised from half an atom of Buckminster fullerene. Properties: •Flexible •Strong •Conduct electricity •High melting point •High boiling point •1 nanometre in diameter (1nm) CNT’s are essentially just half a ‘Bucky ball’.