Biophisical Properties - Lecture -1.pptx
- 1. Biophysical Chemistry Dr. Syed Irfan Raza
- 2. PHYSICOCHEMICAL PRINCIPLES Biophysical chemistry aims to bridge the physical and biological disciplines: in biological systems physical forces and interactions are mediated through molecules, which ultimately determine phenotype. General laws & Principles of Chemistry & Physics are applicable https://www.sciencedirect.com/topics/chemistry/biophysical-chemistry
- 3. C h a p t e r O u t l i n e • Water significance & Its dissociation • Hydrogen Ion concentration & pH • Human Body Buffers • Solution & Types • Colloidal State, Classification & Properties • Diffusion & Osmosis and applications • Donannan Membrane Equilibrium & Applications • Viscosity & Its application • Surface Tension & Its application • Absorption & Adsorption • Isotopes & Radio activity
- 4. Biophysical Properties - Lecture 1 Learning Objectives Students will be able to: • Structure of water and its Chemistry significance • Under stand dissociation of water • Concentration of [H+] & [OH-] in pure water and Kw • pH and its calculation • Define Buffers their composition & types • Explain Mechanism of Buffers
- 6. 65 – 70 % Dipole – Asymmetric structure Hydrogen bonding – phases of H2O • Solid • Liquid • Gas H- Bonding • Viscosity • Surface Tension Significance of Water
- 7. Dissociation of water Pure Water is weak electrolyte Dissociation/ Protolysis is reversible H2O ↔ H+ + OH- / 107 molecules OH- g. mol/L x H+ g. mol/L= kw Kw dependent on temperature & = 0.000000000000001 or 10-14 @ 250 C In Pure Water OH- = H+ = Hence Neutral
- 8. Hydrogen Ion Concentration of water Pure Water: Molecular weight of H2O = (2+16) = 18 1 Liter of water = 1000 grams = 1000/18 = 55.56 moles Hence we can express: 55.56 moles of water contain = 1 H+ & 1OH- pH = H+ ion concentration of any solution
- 9. 1. What is true about 55.56 moles of pure water: a. pH = 1 b. pOH = 1 c. [H+] = 1 d. [OH-] = 7 e. Kw = 107
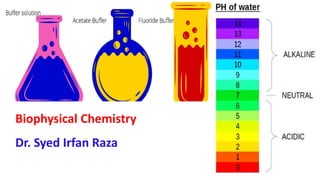
- 11. Biophysical Properties • pH= Hydrogen Ion concentration = [H+] • [H+] expressed in = 0 → 7 → 14 Sorenson – 1909: pH is defined as: The negative logarithm of H+ ion concentration pH = -log [H+]
- 12. Acidic & Basic Solutions
- 13. B u f f e r s pH of a solution can be easily changed by the addition of acids or base