Signal Transduction
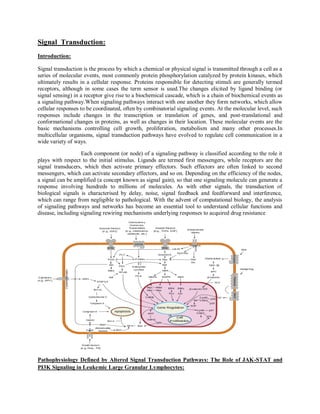
- 1. Signal Transduction: Introduction: Signal transduction is the process by which a chemical or physical signal is transmitted through a cell as a series of molecular events, most commonly protein phosphorylation catalyzed by protein kinases, which ultimately results in a cellular response. Proteins responsible for detecting stimuli are generally termed receptors, although in some cases the term sensor is used.The changes elicited by ligand binding (or signal sensing) in a receptor give rise to a biochemical cascade, which is a chain of biochemical events as a signaling pathway.When signaling pathways interact with one another they form networks, which allow cellular responses to be coordinated, often by combinatorial signaling events. At the molecular level, such responses include changes in the transcription or translation of genes, and post-translational and conformational changes in proteins, as well as changes in their location. These molecular events are the basic mechanisms controlling cell growth, proliferation, metabolism and many other processes.In multicellular organisms, signal transduction pathways have evolved to regulate cell communication in a wide variety of ways. Each component (or node) of a signaling pathway is classified according to the role it plays with respect to the initial stimulus. Ligands are termed first messengers, while receptors are the signal transducers, which then activate primary effectors. Such effectors are often linked to second messengers, which can activate secondary effectors, and so on. Depending on the efficiency of the nodes, a signal can be amplified (a concept known as signal gain), so that one signaling molecule can generate a response involving hundreds to millions of molecules. As with other signals, the transduction of biological signals is characterised by delay, noise, signal feedback and feedforward and interference, which can range from negligible to pathological. With the advent of computational biology, the analysis of signaling pathways and networks has become an essential tool to understand cellular functions and disease, including signaling rewiring mechanisms underlying responses to acquired drug resistance Pathophysiology Defined by Altered Signal Transduction Pathways: The Role of JAK-STAT and PI3K Signaling in Leukemic Large Granular Lymphocytes:
- 2. Signal transduction pathways integrate a variety of micro environmental cues to guide cell function by regulating gene transcription, cell cycle status, growth, and differentiation. It is well established that perturbation of these processes plays a key role in hematologic malignancies including lymphomas and chronic and acute lymphocytic leukemias. Altered intracellular signaling pathways have been proposed to mediate many biological properties of T cell large granular lymphocytic leukemia (T‑LGL), a disorder characterized by a clonal proliferation of CD8 T cells resulting in immune‑mediated cytopenias,most commonly neutropenia. Since T‑LGL offers a unique opportunity to study signal transduction in the pathologic clonal cytotoxic T cell (CTL) compared to normal CTL, we have investigated a potential imbalance in T‑LGL pro‑survival signaling to define the mechanisms underlying the semi‑autonomous proliferation leading to leukemia. Increased activity of the PI3K‑AKT signaling axis in T‑LGL cells appears to operate in conjunction with or parallel to increased STAT3 activation in these cells to inhibit the apoptotic program. Thus, the ability to define pathophysiology at the molecular level opens new avenues for targeted therapeutics. The well‑orchestrated induction of signal transduction pathways defines cellular behavior through complex interactions that are only beginning to be worked out in experimental systems. Despite the daunting task of comprehending all aspects of the entire networked system, focused approach of interrogating specific signaling pathways,depending on the hypothesis being tested can provide useful information regarding pathophysiology and yield potential therapeutic targets. The highly dynamic nature of the immune system, with a multitude of interconnected accessory receptor and cytokine receptor signaling pathways, is an ideal setting to test this premise. Nature of Clonal Expansion of in CTL from T‑LGL and their in Normal Counterparts Within the cell‑mediated arm of the host immune response, cytotoxic CD8 T cells (CTL) have a well‑defined role in targeted killing of malignant and virus‑infected cells. CTL activation is initiated via T cell receptor signaling, and augmented through a multitude of accessory receptor‑ligand signaling systems resulting in the production of cytokines and release of cytotoxic factors. In the waning phase of an immune response, signal transduction pathways are initiated to instruct the vast majority of activated CD8 T cells directed against inciting antigens to undergo apoptosis.Failure of this signaling system due to genetic lesions has been linked to lymphoproliferative disorders.Thus, it is rational to link malfunction of apoptotic mechanisms to autoimmune disease. In certain individuals, a T cell clone may emerge that is largely resistant to the factors driving a return to the homeostatic set point, thus setting the stage for a neoplastic process. T cell large granular lymphocytic leukemia (T‑LGL) is a suitable model to study physiologic and pathophysiologic regulation of CTL clonal expansion and persistence. Transduction in T‑LGL T‑LGL is a clonal lymphoproliferative process of CD8 cytotoxic T cells, often arising in the context of various autoimmune conditions and resembling a natural CTL response to viral infection.A key aspect of this disease is the failure of previously activated cytotoxic T cells to undergo apoptosis, despite appropriate expression of Fas and FasL. However, the T‑LGL cells are relatively insensitive to anti‑Fas or FasL induced apoptosis.A molecular mechanism for apoptotis resistance was recently proposed with the finding that STAT3 is constitutively phosphorylated at tyrosine 705 in T‑LGL, resulting in increased DNA binding activity and transcription of the anti‑apoptotic protein Mcl‑1.14 Since T‑LGL is in large part a disease characterized by failure to undergo homeostatic apoptosis by a single (or few) CTL clones, we considered additional pro‑survival pathways that may be aberrantly activated in these cells and analyzed a possible link between insensitivity to Fas‑induced apoptotic signaling and increased activation of STAT3. The involvement of phosphatidylinositol‑3 kinase (PI3K) signal transduction pathway in the LGL dysregulation was considered likely because of its ability to inhibit Fas signaling15 and its role in regulating STAT3 serine phosphorylation in primary T cells,16 believed to be required for optimal transcriptional activation.We found increased PI3K‑AKT pathway activity in T‑LGL cells compared to T cells from healthy donors, suggesting inappropriate activation of this pro‑survival signaling pathway. A proposed mechanism for JAK‑STAT and PI3K pathway crosstalk
- 3. in T cells involves the ERK mitogen activated protein kinase pathway (MAPK).We have previously shown that optimal phosphorylation of ERK in primary T cells is dependent on the prior activation of the PI3K‑AKT pathway. Thus increased PI3K‑AKT activation may indeed be decreasing the threshold for ERK activation in T‑LGL cells and positively influencing JAK‑STAT signaling . However, it is also possible that these two pathways are acting in parallel to enhance cell survival.A key question that needs to be addressed is how the PI3K‑AKT pathway is aberrantly activated in the T‑LGL cells. A simplistic way of approaching this question is to consider the opposing effects of activating factors that induce pathway activation and inhibitory factors that dampen pathway activation. For instance, LGL patients show a constitutively upregulated production of IL‑18, RANTES, and MIP‑1b, which are known to activate the PI3K pathway.Thus, it is possible that increased PI3K‑AKT pathway activation is drivenin part by increased cytokine receptor signaling. Since our recent publication in Blood describing Src family kinase (SFK) dependent PI3K‑AKT pathway activation in T‑LGL,we have identified potential defects in the negative regulation of this signaling network as well, based on results from gene expression profiling (manuscript submitted). In particular, we have observed decreased expression of three genes in particular that relate to control of SFK activity (PAG1) and PI3K‑AKT activation (PTEN and PIK3R1). Thus, it is possible that the normal “brakes” regulating the pro‑survival signaling network are not functioning optimally . Since signal transduction pathways operate as cascades, it is conceivable that a small perturbation in the proximal part of the pathway, due to slightly decreased inhibition, will be amplified in the downstream elements regulating survival versus apoptosis (such an effect may be further exaggerated in the context of continuous cytokine receptor activation, or persistent antigenic drive, e.g., during an autoimmune process). The role of increased SFK activity in T‑LGL, and its requirement for the increased activity of the PI3K‑AKT pathway, is interesting in light of long‑sought association of T‑LGL with a viral infection.Several viruses have demonstrated an ability to induce PI3K‑AKT. activity in an SFK dependent fashion. This effect was associated with the ability of virus molecular machinery to inhibit host cell apoptosis. While no definitive viral etiology has been linked with T‑LGL, this area is under active investigation. The possibility that SFK activation plays a role in T‑LGL pathogenesis is extended by our recent observation that mRNA expression of phosphoprotein associated with glycolipid‑enriched domains (PAG) is decreased in these cells compared to healthy individuals (unpublished observations). Consequently one could hypothesize that the reduced levels of PAG will diminish the membrane recruitment of Csk, a kinase that associates with PAG and phosphorylates the inhibitory C‑terminal tyrosine in SFKs24 . Thus, the SFK activity in glycolipidenriched domains, also referred to as lipid rafts, of T‑LGL cells would be missing an important inhibitory signal and thus have a lower threshold for activation. Elucidating the relationship of PAG, Csk, and the different phosphorylated forms of SFK (positive regulatory site in the kinase loop domain and negative regulatory Csk‑targeted site near the C‑terminus) in lipid rafts may help to explain the mechanism for the increased SFK activation we see in T‑LGL. In addition, we are investigating the spatial context in which lipid raft associated SFK interact with and positively influence PI3K and/or AKT kinase activity, either directly or via inhibition of PTEN. It is likely that a greater understanding of this networking interaction (SFK pathways interacting with the PI3K pathway) will shed light on the mechanisms by which the anti‑apoptotic PI3K pathway may be regulated in normal and disease states. The decreased PTEN expression in T‑LGL cells, noted above, is an interesting finding in that this is a known mechanism for neoplastic cells to thwart the normal balance of PI3K signaling.In conjunction with any negative regulation by increased SFK activity,25 decreased PTEN could markedly hinder the ability to keep PI3K dependent pathways in check. A less obvious mechanism for a relative increase in PI3K activity might be decreased expression of p85a (PIK3R1), a regulatory subunit of class IA PI3K enzymes. While the class I PI3K enzymes function as heterodimers of regulatory and catalytic subunits, it has been shown that decreased expression of the regulatory subunit can actually increase the activation of PI3K.For example, in wild‑type fibroblasts, the
- 4. p85 subunit was more abundant than p110, leading to competition between the p85 monomer and the p85‑p110 dimer for receptor binding sites and diminished PI3K signaling. When p85 was reduced by genetic means, PI3K signaling was enhanced, suggesting that a new balance between the lone regulatory subunit and the p85/p110 dimer had been reached.Consequently, aberrant activation of PI3K‑AKT pathway in T‑LGL cells in the context of decreased PIK3R1 expression, may suggest yet another defective negative regulatory pathway, allowing pro‑survival signaling to continue with less inhibitory input. We are currently studying the potential crosstalk between the PI3K‑AKT pathway and the JAK‑STAT pathway in T‑LGL. While the published role of ERK as an intermediary in this regard is appealing, the role of STAT3 (S727) phosphorylation in CTL activation needs to be better defined. Notably, in other tissues, (e.g., mammary epithelial cells), the relationship between STAT3 activation and PI3K‑AKT is quite different: mammary gland involution is dependent on STAT3 mediated apoptosis, due in part to PI3K‑AKT inhibition. This is in stark contrast to T‑LGL cells, in which STAT3 drives transcription of the anti‑apoptotic protein Mcl‑1, highlighting how signaling pathways interact to direct effector function in a cell-specific manner.14 Inhibition of the JAK‑STAT pathway via the tyrphostin AG490 resulted in apoptosis of the T‑LGL cells, illustrating the importance of the cellular context in which signal transduction pathways are networked together. Thus, evolutionary processes have selected for modular signaling pathways that can be assembled in unique spatial and temporal configurations to achieve cell and tissue‑type dependent responses. Aberrant survival signaling in T‑LGL cells serve an important reminder that pathologic activation of signal transduction pathways is not necessarily due to overexpression of signaling components, such as that classically seen in Her2/Neu amplification in breast cancer,or to genetic lesions creating increased intrinsic enzymatic activity, such as the recently described JAK2 V617F mutation in myeloproliferative disorders. We believe that it is important to consider a loss of the inhibitory factors that are required to maintain a dynamic homeostasis. Decreased expression of adaptor molecules that possess no intrinsic enzymatic activity themselves, such as PAG, may be easily overlooked, but can play an important role in regulating the overall signaling network. Decreased expression of the PTEN protein, or a mutation resulting in decreased phosphatase activity is an obvious clue that the PI3K‑AKT pathway may lack an important regulatory input. However, downmodulation of the p85a subunit of the PI3K heterodimeric enzyme may result in a decreased ratio of free p85aa to p85a in complex with the catalytic subunit. This subtle loss of PI3K regulation at the level of initial recruitment to activated receptors may have profound functional consequences. Conclusion: Signal transduction, and ultimately signaling networks, becomes more sophisticated, so to must our appreciation for the myriad controls that operate on many levels. The fact that signaling pathways may be differentially constructed and regulated in various tissue types may at first glance seem to defy our ability to understand these pathways in any meaningful detail. However, we believe that this provides an excellent opportunity to observe how different modular signaling components can be used to integrate common signals into unique cellular and tissue responses,and ultimately delineate novel targets for emerging therapies.
- 5. Signal transduction by the epidermal growth factor receptor after functional desensitization of the receptor serine protein kinase activity: Introduction: Previous work identified a protein kinase activity that phosphorylates the epidermal growth factor (EGF) receptor at Thr"'. An assay for this protein kinase activity present in homogenates prepared from A431 human epidermoid carcinoma cells was developed using a synthetic peptide substrate corresponding to residues 663-681 of the EGF receptor (peptide T669). Here we report that a greater initial rate of T669 phosphorylation was observed in experiments using homogenates prepared from EGF- or phorbol ester-treated cells compared with control cells. EGF and 4p-phorbol 12- myristate 13-acetate (PMA) caused a 6-fold and a 2-fold increase in protein kinase activity, respectively. A kinetic analysis of T669 phosphorylation demonstrated that the increase in protein kinase activity observed was accounted for by an increase in Vmax. To examine the interaction between protein kinase C and signal transduction by the EGF receptor, the effect of pretreatment of cells with PMA on the subsequent response to EGF was investigated. Treatment of cells with PMA caused >90% inhibition of the EGF-stimulated tyrosine phosphorylation of the EGF receptor and abolished the EGFstimulated formation of soluble inositol
- 6. phosphates. In contrast, PMA was not observed to inhibit the stimulation of T669 protein kinase activity caused by EGF. Thus, the apparent functional desensitization of the EGF receptor caused by PMA does not inhibit signal transduction mediated by the T669 protein kinase. Our results demonstrate that EGF receptor transmodulation alters the pattern of signal-transduction pathways that are utilized by the EGF receptor. Transmembrane signaling by the epidermal growth factor (EGF) receptor is acutely regulated by protein kinase C. Activation of protein kinase C by treatment of cells with phorbol ester tumor promoters causes an inhibition of the high-affinity binding of EGF to cell surface receptors and causes an inhibition of the receptor tyrosine protein kinase activity . This process of acute regulation of EGF receptor function has been termed transmodulation . The molecular mechanism that accounts for transmodulation and the role of transmodulation in signal transduction by the EGF receptor are not understood. However, signaling by the transmodulated EGF receptor has been studied in detail. It has been demonstrated that EGF-stimulated phosphatidylinositol turnover and the generation of soluble inositol phosphates are inhibited by incubation of cells with phorbol ester . Phorbol ester also inhibits the increase in cytosolic Ca2+ caused by EGF without affecting basal cytosolic Ca2+ levels . Furthermore, the EGF-stimulated Na+/H+ exchange across the plasma membrane is inhibited by phorbol ester . Together, these data indicate that protein kinase C causes an acute desensitization of the function of the EGF receptor. Recently we described a protein kinase activity in cell homogenates that phosphorylates the EGF receptor at threonine- 669 . An assay for this protein kinase activity was developed using a synthetic peptide substrate corresponding to residues 663-681 of the EGF receptor (peptide T669). Treatment of cells with EGF caused a marked increase in the level of T669 protein kinase activity detected compared with control cells . The stimulation of T669 protein kinase activity caused by EGF indicates that this enzyme may have an important function in signal transduction. The purpose of the experiments described in this report was to examine the role of protein kinase C in the regulation of T669 protein kinase activity by EGF. Conclusion: Transmodulation ofthe EGF receptor has been reported to cause the desensitization of EGF receptor function . The observation reported here that the transmodulated EGF receptor is capable of signal transduction (mediated by the T669 protein kinase) indicates that transmodulation does not result in the complete desensitization of EGF receptor function. Instead, the results of this study demonstrate that EGF receptor transmodulation changes the pattern of signal-transduction pathways that are utilized by the EGF receptor. This conclusion may be physiologically significant because EGF receptor transmodulation caused by platelet- derived growth factor results in the synergistic stimulation of the growth of cells cultured with EGF.
- 8. G protein–coupled receptors: 1.G protein–coupled receptors (GPCRs) are a family of integral transmembrane proteins that possess seven transmembrane domains and are linked to a heterotrimeric G protein. With nearly 800 members, this is the largest family of membrane proteins and receptors in mammals. Counting all animal species, they add up to over 5000. 2.Mammalian GPCRs are classified into 5 major families: rhodopsin-like, secretin-like, metabotropic glutamate, adhesion and frizzled/smoothened, with a few GPCR groups being difficult to classify due to low sequence similarity, e.g. vomeronasal receptors.Other classes exist in eukaryotes, such as the Dictyostelium cyclic AMP receptors and fungal mating pheromone receptors.Signal transduction by a GPCR begins with an inactive G protein coupled to the receptor; the G protein exists as a heterotrimer consisting of Gα, Gβ, and Gγ subunits. 3.Once the GPCR recognizes a ligand, the conformation of the receptor changes to activate the G protein, causing Gα to bind a molecule of GTP and dissociate from the other two G-protein subunits. The dissociation exposes sites on the subunits that can interact with other molecules. 4.The activated G protein subunits detach from the receptor and initiate signaling from many downstream effector proteins such as phospholipases and ion channels, the latter permitting the release of second messenger molecules.
- 9. 5.The total strength of signal amplification by a GPCR is determined by the lifetimes of the ligand- receptor complex and receptor-effector protein complex and the deactivation time of the activated receptor and effectors through intrinsic enzymatic activity; e.g. via protein kinase phosphorylation or b- arrestin-dependent internalization.A study was conducted where a point mutation was inserted into the gene encoding the chemokine receptor CXCR2; mutated cells underwent a malignant transformation due to the expression of CXCR2 in an active conformation despite the absence of chemokine-binding. This meant that chemokine receptors can contribute to cancer development. Tyrosine, Ser/Thr and Histidine-specific protein kinases: 1.Receptor tyrosine kinases (RTKs) are transmembrane proteins with an intracellular kinase domain and an extracellular domain that binds ligands; examples include growth factor receptors such as the insulin receptor.To perform signal transduction, RTKs need to form dimers in the plasma membrane;the dimer is stabilized by ligands binding to the receptor. 2.The interaction between the cytoplasmic domains stimulates the autophosphorylation of tyrosine residues within the intracellular kinase domains of the RTKs, causing conformational changes. Subsequent to this, the receptors' kinase domains are activated, initiating phosphorylation signaling cascades of downstream cytoplasmic molecules that facilitate various cellular processes such as cell differentiation and metabolism. 3.Many Ser/Thr and dual-specificity protein kinases are important for signal transduction, either acting downstream of [receptor tyrosine kinases], or as membrane-embedded or cell-soluble versions in their own right. The process of signal transduction involves around 560 known protein kinases and pseudokinases, encoded by the human kinome 4.As is the case with GPCRs, proteins that bind GTP play a major role in signal transduction from the activated RTK into the cell. In this case, the G proteins are members of the Ras, Rho, and Raf families, referred to collectively as small G proteins. They act as molecular switches usually tethered to membranes by isoprenyl groups linked to their carboxyl ends. 5.Upon activation, they assign proteins to specific membrane subdomains where they participate in signaling. Activated RTKs in turn activate small G proteins that activate guanine nucleotide exchange factors such as SOS1. Once activated, these exchange factors can activate more small G proteins, thus amplifying the receptor's initial signal. 6. The mutation of certain RTK genes, as with that of GPCRs, can result in the expression of receptors that exist in a constitutively activated state; such mutated genes may act as oncogenes.Histidine-specific protein kinases are structurally distinct from other protein kinases and are found in prokaryotes, fungi, and plants as part of a two-component signal transduction mechanism: a phosphate group from ATP is first added to a histidine residue within the kinase, then transferred to an aspartate residue on a receiver domain on a different protein or the kinase itself, thus activating the aspartate residue.
