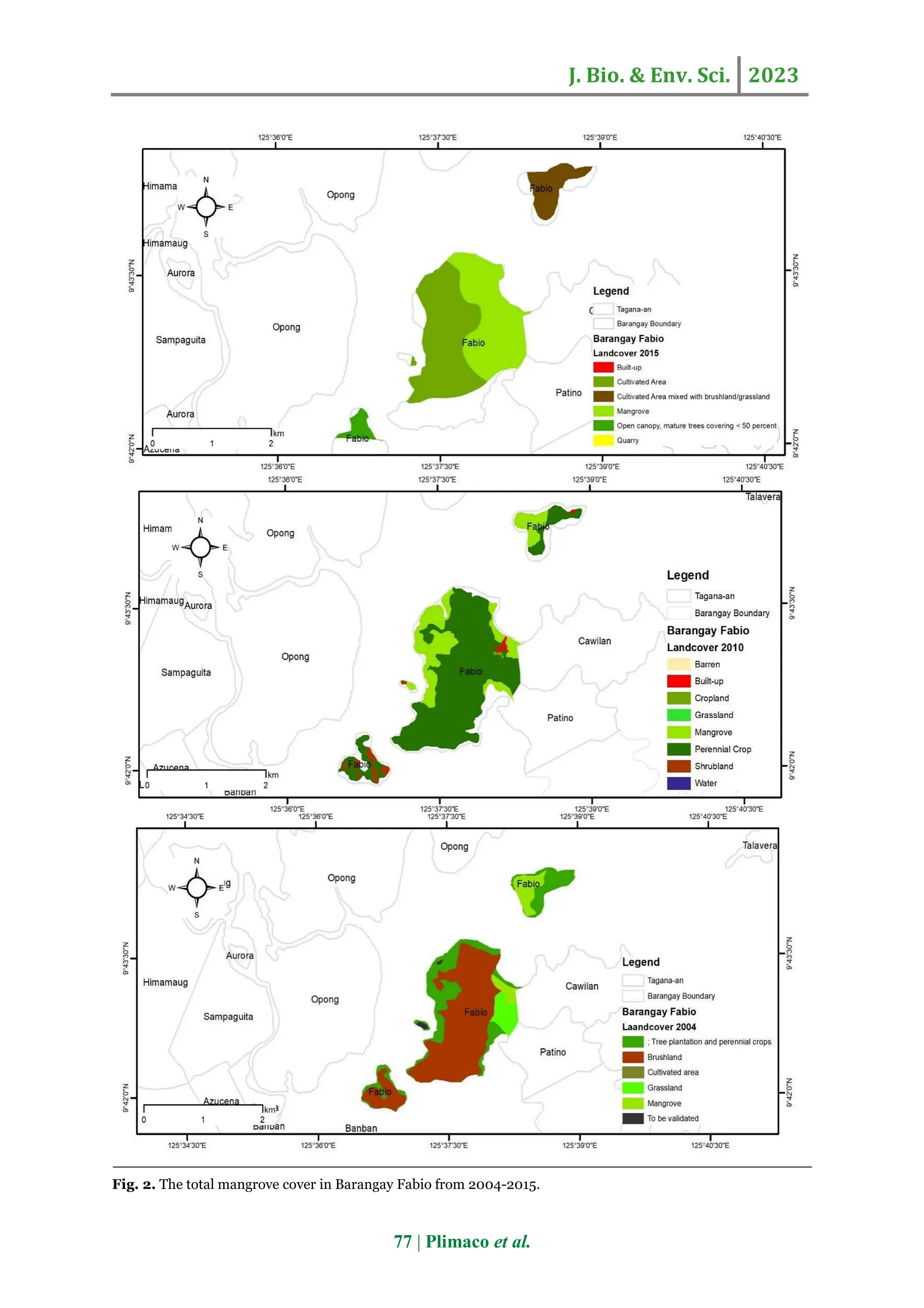
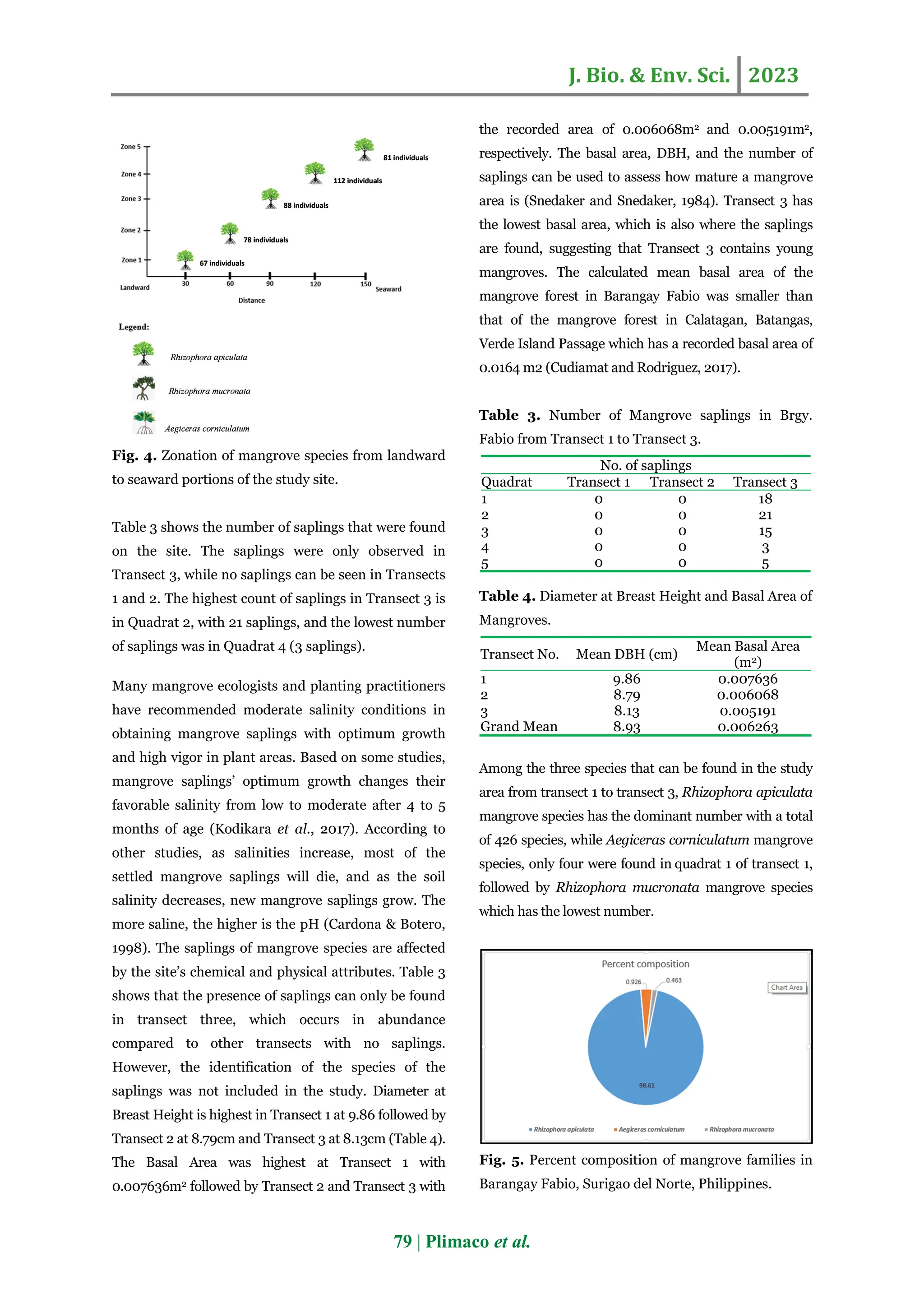
The study assessed the species composition, diversity, and community structure of mangrove forests in Barangay Fabio, Surigao del Norte, Philippines, revealing low diversity with only three mangrove species belonging to two families. Rhizophora apiculata was found to be the most abundant species, highlighting the area’s ecological vulnerability due to declining mangrove coverage caused by anthropogenic activities. Effective conservation programs are recommended to enhance mangrove preservation in the region.