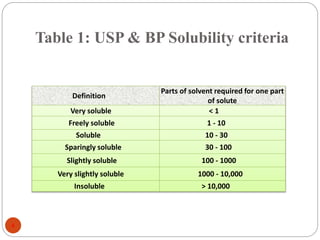
This document discusses various techniques for improving the solubility of poorly soluble drugs, which is important for enhancing their bioavailability. It describes 15 techniques in detail, including particle size reduction through micronization and nanosuspensions, solubilization using hydrotropy, cosolvency and surfactants, and formation of solid dispersions and complexes. Other techniques discussed are pH adjustment, high pressure homogenization, supercritical fluid processing, and sonocrystallization. The document provides examples and explanations of how each technique can increase a drug's solubility.