Scott Doney's Ocean Acidification presentation, April 2013 Hourglass Brasserie
•Download as PPTX, PDF•
2 likes•483 views
Dr. Scott Doney from Woods Hole Oceanographic Institution joined a group of guests at the Hourglass Brasserie, Bristol RI, in April 2013 to offer some thoughts on the effects of ocean acidification on New England's treasured seafood.
Report
Share
Report
Share
Recommended
Ocean acidification: the process, the future, and the past

This document discusses ocean acidification, including its causes from increased carbon dioxide in the atmosphere, its impacts such as negative effects on coral reefs and calcifying organisms, and evidence that it has occurred before in the past due to natural processes. The future remains uncertain but ocean acidification is projected to continue increasing stresses on marine ecosystems when combined with global warming. Some research also indicates acidification may impact organisms and ecosystems differently depending on location.
New meg chadsey northwest school presentation with credits dec27

This document summarizes ocean acidification, its causes and impacts. It begins by explaining that as the ocean absorbs increasing amounts of carbon dioxide from the atmosphere, the pH of seawater decreases, making it more acidic. This phenomenon is known as ocean acidification. It then discusses how ocean acidification particularly impacts the Northwest region of the United States by discussing the factors that exacerbate the problem locally. The document also summarizes the economic and cultural impacts of ocean acidification for Washington state in terms of fisheries, shellfish industries, tribal communities and more. It concludes by outlining recommendations from Washington's Blue Ribbon Panel on Ocean Acidification and actions the state legislature has taken to fund the panel's proposed response strategies
Corals and global warming: the Mediterranean VS the Red Sea

The project aims to create a predictive model of coral survival and reef community structure changes in the Mediterranean and Red Sea under climate change scenarios over the next century. It will examine the effects of temperature warming and ocean acidification on corals and calcifying organisms. The interdisciplinary project brings together experts from various fields like photobiology, physiology, crystallography and population dynamics to study these impacts.
The Upwell #Acidinar: Ocean Acidification Through the Eyes of the Internet

From the twisted minds who brought you the Upwell Sharkinar, comes the latest in our infamous webinar-inar series. The Acidinar is about using the internet for ocean acidification communications, including what can be done about it.
If you talk about acidification online, or if you're acidification-curious, check out the Acidinar and join our merry band of activists, scientists, bloggers, journalists, super-tweeters, and nonprofits to discuss how we can change the online conversation about ocean acidification in the best way, together.
The Acidinar was held on Wednesday, February 4th, at 11am pacific / 2pm eastern.
A video recording and related resources are available at http://upwell.us/oceanacidification
During the Acidinar we:
• Shared findings from Upwell’s new report, Ocean Acidification: The State of the Online Conversation (http://upwell.us/oceanacidification)
• Answered your questions and discussed your takeaways, with an eye toward mutual communications support
• Provided tips for increasing the volume and engagement level of ocean acidification online mentions
SAGB2013 Dr Ed Pope/Dr Frances Hopkins (UK Ocean Acidification Consortium)

The document summarizes a UK Ocean Acidification Research Programme that studied the effects of ocean acidification on marine ecology, biogeochemistry, and shellfish. It was a 4-year, £12M project involving 150 scientists from 23 institutions. The program found measurable increases in ocean acidity and decreases in carbonate ions due to rising atmospheric CO2 levels. Studies on shellfish showed their early life stages are more sensitive to acidification. However, adequate nutrition and selective breeding could make populations more tolerant. The research provided a baseline to assess future climate change projections and their economic impacts on UK aquaculture.
Ocean acidification

Ocean acidification is a term used to describe the changes in the chemistry of the Earth’s ocean i.e. ongoing decrease in the pH and increase in acidity caused by the uptake of anthropogenic carbon dioxide from the atmosphere causing major problems for the coral reefs and other organisms.
ocean acidification

Ocean acidification is caused by increasing carbon dioxide levels in the oceans due to human emissions since the Industrial Revolution. As CO2 is absorbed by seawater, chemical reactions occur that reduce seawater pH and the concentration of carbonate ions. This process is known as ocean acidification and impacts marine life by making it difficult for calcifying organisms like oysters, corals, and plankton to form their shells and skeletons. While some algae may benefit from higher CO2 levels, most marine species face threats of thinner shells, lower survival rates, and lower population growth under increasingly acidic conditions. Options to mitigate ocean acidification include reducing CO2 emissions, allowing species to adapt or relocate, or developing carbon capture
Ocean Acidification: Cause, Impact and mitigation

Ocean Acidification and the battle for Carbonate.
In this presentation the points covered are detailed briefing of ocean acidification, its causes, its impact on marine ecosystems and measures to mitigate this.
Recommended
Ocean acidification: the process, the future, and the past

This document discusses ocean acidification, including its causes from increased carbon dioxide in the atmosphere, its impacts such as negative effects on coral reefs and calcifying organisms, and evidence that it has occurred before in the past due to natural processes. The future remains uncertain but ocean acidification is projected to continue increasing stresses on marine ecosystems when combined with global warming. Some research also indicates acidification may impact organisms and ecosystems differently depending on location.
New meg chadsey northwest school presentation with credits dec27

This document summarizes ocean acidification, its causes and impacts. It begins by explaining that as the ocean absorbs increasing amounts of carbon dioxide from the atmosphere, the pH of seawater decreases, making it more acidic. This phenomenon is known as ocean acidification. It then discusses how ocean acidification particularly impacts the Northwest region of the United States by discussing the factors that exacerbate the problem locally. The document also summarizes the economic and cultural impacts of ocean acidification for Washington state in terms of fisheries, shellfish industries, tribal communities and more. It concludes by outlining recommendations from Washington's Blue Ribbon Panel on Ocean Acidification and actions the state legislature has taken to fund the panel's proposed response strategies
Corals and global warming: the Mediterranean VS the Red Sea

The project aims to create a predictive model of coral survival and reef community structure changes in the Mediterranean and Red Sea under climate change scenarios over the next century. It will examine the effects of temperature warming and ocean acidification on corals and calcifying organisms. The interdisciplinary project brings together experts from various fields like photobiology, physiology, crystallography and population dynamics to study these impacts.
The Upwell #Acidinar: Ocean Acidification Through the Eyes of the Internet

From the twisted minds who brought you the Upwell Sharkinar, comes the latest in our infamous webinar-inar series. The Acidinar is about using the internet for ocean acidification communications, including what can be done about it.
If you talk about acidification online, or if you're acidification-curious, check out the Acidinar and join our merry band of activists, scientists, bloggers, journalists, super-tweeters, and nonprofits to discuss how we can change the online conversation about ocean acidification in the best way, together.
The Acidinar was held on Wednesday, February 4th, at 11am pacific / 2pm eastern.
A video recording and related resources are available at http://upwell.us/oceanacidification
During the Acidinar we:
• Shared findings from Upwell’s new report, Ocean Acidification: The State of the Online Conversation (http://upwell.us/oceanacidification)
• Answered your questions and discussed your takeaways, with an eye toward mutual communications support
• Provided tips for increasing the volume and engagement level of ocean acidification online mentions
SAGB2013 Dr Ed Pope/Dr Frances Hopkins (UK Ocean Acidification Consortium)

The document summarizes a UK Ocean Acidification Research Programme that studied the effects of ocean acidification on marine ecology, biogeochemistry, and shellfish. It was a 4-year, £12M project involving 150 scientists from 23 institutions. The program found measurable increases in ocean acidity and decreases in carbonate ions due to rising atmospheric CO2 levels. Studies on shellfish showed their early life stages are more sensitive to acidification. However, adequate nutrition and selective breeding could make populations more tolerant. The research provided a baseline to assess future climate change projections and their economic impacts on UK aquaculture.
Ocean acidification

Ocean acidification is a term used to describe the changes in the chemistry of the Earth’s ocean i.e. ongoing decrease in the pH and increase in acidity caused by the uptake of anthropogenic carbon dioxide from the atmosphere causing major problems for the coral reefs and other organisms.
ocean acidification

Ocean acidification is caused by increasing carbon dioxide levels in the oceans due to human emissions since the Industrial Revolution. As CO2 is absorbed by seawater, chemical reactions occur that reduce seawater pH and the concentration of carbonate ions. This process is known as ocean acidification and impacts marine life by making it difficult for calcifying organisms like oysters, corals, and plankton to form their shells and skeletons. While some algae may benefit from higher CO2 levels, most marine species face threats of thinner shells, lower survival rates, and lower population growth under increasingly acidic conditions. Options to mitigate ocean acidification include reducing CO2 emissions, allowing species to adapt or relocate, or developing carbon capture
Ocean Acidification: Cause, Impact and mitigation

Ocean Acidification and the battle for Carbonate.
In this presentation the points covered are detailed briefing of ocean acidification, its causes, its impact on marine ecosystems and measures to mitigate this.
Ocean acidification

The oceans are getting acidified! How? Why? What can we do?
For answers... Check the Presentation out
(Just a bit more informative than my previous one)
Ocean accidification inna_tarasyan

The document discusses ocean acidification which is caused by the uptake of anthropogenic CO2 by the oceans. Approximately 45% of anthropogenic CO2 emissions between 2000-2010 were absorbed by the oceans, causing the pH of ocean water to decline. This impacts marine life such as coral reefs and organisms with calcareous skeletons, as it decreases their growth rates. The document recommends supporting policies to reduce carbon emissions, conserving coastal ecosystems, and supporting marine conservation to help oceans become more resilient to stressful conditions caused by acidification.
Richard Feeley presentation on ocean acidification

The document discusses ocean acidification due to increasing atmospheric CO2. It outlines the impacts on marine organisms like coccolithophores, foraminifera, pteropods, mussels and oysters based on experimental studies. These include reduced calcification rates and dissolution of shells. Food webs may be affected if prey types of animals like juvenile salmon are impacted. Monitoring of trends and ecosystem responses is needed along with developing adaptation strategies.
Ocean Acidification and Ocean Noise

Ocean acidification is caused by higher levels of carbon dioxide in the atmosphere being absorbed by the ocean, which increases the acidity of ocean water over time. The pH of ocean water has dropped from around 8.2 to 8.1 and is projected to decrease by 0.3 to 0.4 units over the next century. As the pH of the ocean decreases, it absorbs more CO2 from the atmosphere. This interacts with water molecules to form carbonic acid, lowering pH further. Additionally, ocean acidification may amplify ocean noise levels by around 10% and extend the range of underwater sounds by about 500 kilometers. This can negatively impact marine species by disrupting communication, increasing stress levels, and potentially causing hearing
Ocean Acidification

The ocean absorbs about one-third of carbon dioxide emissions from human activities. This uptake benefits society by slowing climate change but causes ocean acidification as CO2 reacts with seawater. Ocean acidification threatens marine organisms that build shells and skeletons, and could disrupt marine food webs and ecosystems. Future projections estimate the oceans will become 150% more acidic by 2100 if emissions continue unabated, reaching levels not seen for over 20 million years. Strengthening the science of ocean acidification impacts is urgently needed to inform decision making.
Ocean Acidification

The document discusses ocean acidification, which is the ongoing decrease in ocean pH caused by absorbing CO2 from the atmosphere. This absorption has lowered ocean pH by 0.1 units since the pre-industrial period. Ocean acidification affects organisms that rely on calcium carbonate to build shells and skeletons, as acidity decreases availability of carbonate ions. It also impacts metabolism, photosynthesis, nutrient absorption and more. Effects vary by ecosystem but tropical coral reefs, polar regions, and deep sea corals are threatened by slowed growth and structural damage if acidification continues unchecked. Mitigation requires reducing CO2 emissions and improving ocean health.
Ocean acidification

Ocean Acidification atau Pengasaman samudra adalah salah satu dampak peningkatan gas rumah kaca yang berupa CO2 dimana terjadi penurunan pH perairan akibat semakin banyaknya gas CO2 yang diserap laut/perairan
Ocean acidification will-the_reef_survive-non-narrated[1]![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
The burning of fossil fuels releases 11 billion tons of carbon dioxide into the atmosphere each year, which dissolves into the ocean and causes ocean acidification. Ocean acidification threatens shell-forming organisms as a small pH change equals a large acidity change, reducing the concentration of carbonate ions needed by corals and plankton for shell formation. Experiments show yeast producing carbon dioxide that dissolves in water, lowering the pH and demonstrating how ocean acidification occurs.
Ocean acidification

The document discusses the effects of increasing carbon dioxide levels in the atmosphere and oceans. It notes that the ocean absorbs one third of excess CO2 in the atmosphere, and that CO2 concentrations have risen from 280 to 387 parts per million currently and are projected to reach 450 to 550 ppm by volume. It also explains that the ocean is becoming more acidic as it absorbs more CO2, with the pH dropping by 0.1 currently and projected to decrease by 0.3 to 0.7 additional units by 2300, putting coral, clams, and plankton at risk.
Ocean Acidification

Ocean acidification is caused by carbon dioxide from the atmosphere being absorbed by the oceans, which increases acidity levels. Before a few decades, ocean water was basic but acidity has been rising. This poses threats such as damage to marine life, food webs, and ocean goods/services that rely on calcification. Programs are working to increase awareness and research mitigation strategies, though no large-scale reductions have occurred yet. Individual actions like reducing carbon footprints, planting trees, and minimizing vehicle use can help address the problem.
C5.01: Ocean acidification and seawater carbon chemistry of the Great Barrier...

C5.01: Ocean acidification and seawater carbon chemistry of the Great Barrier...Blue Planet Symposium
This document summarizes research on ocean acidification and carbonate chemistry in the Great Barrier Reef. The research aims to determine how exposure to ocean acidification is changing on the reefs and identify drivers of variability. Observations from sites along the Great Barrier Reef and models show that seasonal changes in carbonate chemistry are linked to a winter source of CO2 on the Great Barrier Reef shelf. Coastal inputs also influence carbonate chemistry. The research uses data from ship surveys, moorings and sampling to improve biogeochemical models of the region from individual reef to whole Great Barrier Reef scales.Ocean Acidification Summary for Policymakers (2013)

The Ocean Acidification Summary for Policymakers was published in 2013. It summarises key findings from the Ocean in a High CO2 World Symposium, which is sponsored by the International Geosphere-Biosphere Programme, the Intergovernmental Oceanographic Commission of UNESCO and the Scientific Committee on Oceanic Research.
For more information
http://ocean-acidification.net/
Summary for Policymakers
http://www.igbp.net/publications/summariesforpolicymakers/summariesforpolicymakers/oceanacidificationsummaryforpolicymakers2013.5.30566fc6142425d6c9111f4.html
Ocean acidification & its prevention

Ocean acidification is caused by carbon dioxide emissions dissolving in oceans and reacting to form carbonic acid, increasing hydrogen ions and acidity. This impacts marine organisms like depressing immune responses in mussels and causing coral bleaching. The biggest contributors are livestock farming and fossil fuel use, so reducing meat consumption, using less energy, driving less, and choosing renewable energy can help prevent further acidification. Making sustainable consumer choices regarding plastics, water usage, and manufactured goods can also reduce the carbon footprint driving ocean acidification.
Ocean Acidification

This is a small presentation on ocean acidification.It is a compilation of all materials(including present information) I collected related to it, any new information beside this or concerning it please comment.
Ocean acidification __methane

Conclusion
While methane may be broken down into a less harmful form, when methane is oxidized the problem doesn't simply go away. Methane oxidizes into carbon dioxide both in seawater and in the atmosphere, adding to the carbon dioxide levels in both these realms. As we have seen, carbon dioxide in seawater contributes to ocean acidity, while atmospheric carbon dioxide traps heat and promotes warming – though not quite to the extent that methane would – and as atmospheric CO2 levels rise, so too will the CO2 that is absorbed into the ocean. It is a classic feedback loop that feeds into itself, having the potential to accelerate global warming and climate change to a tipping point – the point of no return.
Ocean Acidification Expert Forum Program

This document provides information about an Ocean Acidification Expert Forum held in February 2015 at the Victoria Conference Centre. It includes the agenda, speaker biographies, and context about ocean acidification. The two-day forum brought together international experts to review research on ocean acidification, identify key research needs for Canada, and establish a way forward for a coordinated Canadian research effort. The goal was to produce a white paper to guide research across sectors within Canada and internationally.
Paper discussion Fabry et al. 2008

1) Increased atmospheric CO2 is being absorbed by the oceans, raising CO2 levels and lowering pH, a process known as ocean acidification.
2) This affects calcifying organisms by decreasing carbonate ion concentrations needed to form calcium carbonate shells and skeletons. It impacts organisms' ability to calcify and causes dissolution of shells.
3) Pteropods, an important food source for salmon and other species, are especially vulnerable as they secrete the more soluble form of calcium carbonate, aragonite. Ocean acidification is restricting their habitat distribution.
Hypoxia and Ocean Acidification project

Dr. Francis Chan's 2012-2014 Oregon Sea Grant-supported project, "Understanding, Forecasting and Communicating the Linkages Between Hypoxia and Ocean Acidification in Oregon's Coastal Ocean"
Ocean acidification

The ocean covers two thirds of the planet and provides half of the oxygen and 20% of the protein for the world's population. However, ocean pollution from human activities is causing problems like ocean acidification. Ocean acidification occurs as the ocean absorbs 25% of the carbon dioxide emitted, which causes the pH of the ocean to decrease as carbon dioxide dissolves in seawater. This chemical change affects over 25% of marine species by decreasing the carbonate ions that many shellfish and corals use to build their shells, putting them at risk of dissolving. To slow ocean acidification and its impacts, carbon dioxide emissions must be reduced.
Climate change

Climate change is caused by human activity that releases heat-trapping greenhouse gases like carbon dioxide and methane. Greenhouse gas levels are the highest they've been in 800,000 years and global temperatures are rising twice as fast as 50 years ago. Effects of climate change include rising temperatures, sea levels and droughts/heat waves that can worsen wildfires. Reducing fossil fuel use and increasing renewable energy and sustainability can help mitigate and adapt to climate change to reduce health impacts.
Discussant: Corinne Le Quéré, Director, Tyndall Centre for Climate Change Res...

The document discusses several emerging issues impacting open oceans from a science perspective, including:
1) Surface salinity and temperature trends from 1950-2011 show changes due to climate change.
2) Ocean acidification has increased since 1979-2007 due to rising atmospheric CO2 levels.
3) Oxygen levels in oceans' top 1000 meters have declined between the 1990s and 1970s.
Peter Ridd

The document discusses potential threats to the Great Barrier Reef, including increased sediment and nutrient levels from agriculture as well as impacts from global warming. While these threats have concerned some, the author argues that the evidence shows the reef is in good condition overall and that the threats have been exaggerated. The author also expresses concerns about bias and lack of open debate in the scientific process around the reef's conservation.
More Related Content
What's hot
Ocean acidification

The oceans are getting acidified! How? Why? What can we do?
For answers... Check the Presentation out
(Just a bit more informative than my previous one)
Ocean accidification inna_tarasyan

The document discusses ocean acidification which is caused by the uptake of anthropogenic CO2 by the oceans. Approximately 45% of anthropogenic CO2 emissions between 2000-2010 were absorbed by the oceans, causing the pH of ocean water to decline. This impacts marine life such as coral reefs and organisms with calcareous skeletons, as it decreases their growth rates. The document recommends supporting policies to reduce carbon emissions, conserving coastal ecosystems, and supporting marine conservation to help oceans become more resilient to stressful conditions caused by acidification.
Richard Feeley presentation on ocean acidification

The document discusses ocean acidification due to increasing atmospheric CO2. It outlines the impacts on marine organisms like coccolithophores, foraminifera, pteropods, mussels and oysters based on experimental studies. These include reduced calcification rates and dissolution of shells. Food webs may be affected if prey types of animals like juvenile salmon are impacted. Monitoring of trends and ecosystem responses is needed along with developing adaptation strategies.
Ocean Acidification and Ocean Noise

Ocean acidification is caused by higher levels of carbon dioxide in the atmosphere being absorbed by the ocean, which increases the acidity of ocean water over time. The pH of ocean water has dropped from around 8.2 to 8.1 and is projected to decrease by 0.3 to 0.4 units over the next century. As the pH of the ocean decreases, it absorbs more CO2 from the atmosphere. This interacts with water molecules to form carbonic acid, lowering pH further. Additionally, ocean acidification may amplify ocean noise levels by around 10% and extend the range of underwater sounds by about 500 kilometers. This can negatively impact marine species by disrupting communication, increasing stress levels, and potentially causing hearing
Ocean Acidification

The ocean absorbs about one-third of carbon dioxide emissions from human activities. This uptake benefits society by slowing climate change but causes ocean acidification as CO2 reacts with seawater. Ocean acidification threatens marine organisms that build shells and skeletons, and could disrupt marine food webs and ecosystems. Future projections estimate the oceans will become 150% more acidic by 2100 if emissions continue unabated, reaching levels not seen for over 20 million years. Strengthening the science of ocean acidification impacts is urgently needed to inform decision making.
Ocean Acidification

The document discusses ocean acidification, which is the ongoing decrease in ocean pH caused by absorbing CO2 from the atmosphere. This absorption has lowered ocean pH by 0.1 units since the pre-industrial period. Ocean acidification affects organisms that rely on calcium carbonate to build shells and skeletons, as acidity decreases availability of carbonate ions. It also impacts metabolism, photosynthesis, nutrient absorption and more. Effects vary by ecosystem but tropical coral reefs, polar regions, and deep sea corals are threatened by slowed growth and structural damage if acidification continues unchecked. Mitigation requires reducing CO2 emissions and improving ocean health.
Ocean acidification

Ocean Acidification atau Pengasaman samudra adalah salah satu dampak peningkatan gas rumah kaca yang berupa CO2 dimana terjadi penurunan pH perairan akibat semakin banyaknya gas CO2 yang diserap laut/perairan
Ocean acidification will-the_reef_survive-non-narrated[1]![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
The burning of fossil fuels releases 11 billion tons of carbon dioxide into the atmosphere each year, which dissolves into the ocean and causes ocean acidification. Ocean acidification threatens shell-forming organisms as a small pH change equals a large acidity change, reducing the concentration of carbonate ions needed by corals and plankton for shell formation. Experiments show yeast producing carbon dioxide that dissolves in water, lowering the pH and demonstrating how ocean acidification occurs.
Ocean acidification

The document discusses the effects of increasing carbon dioxide levels in the atmosphere and oceans. It notes that the ocean absorbs one third of excess CO2 in the atmosphere, and that CO2 concentrations have risen from 280 to 387 parts per million currently and are projected to reach 450 to 550 ppm by volume. It also explains that the ocean is becoming more acidic as it absorbs more CO2, with the pH dropping by 0.1 currently and projected to decrease by 0.3 to 0.7 additional units by 2300, putting coral, clams, and plankton at risk.
Ocean Acidification

Ocean acidification is caused by carbon dioxide from the atmosphere being absorbed by the oceans, which increases acidity levels. Before a few decades, ocean water was basic but acidity has been rising. This poses threats such as damage to marine life, food webs, and ocean goods/services that rely on calcification. Programs are working to increase awareness and research mitigation strategies, though no large-scale reductions have occurred yet. Individual actions like reducing carbon footprints, planting trees, and minimizing vehicle use can help address the problem.
C5.01: Ocean acidification and seawater carbon chemistry of the Great Barrier...

C5.01: Ocean acidification and seawater carbon chemistry of the Great Barrier...Blue Planet Symposium
This document summarizes research on ocean acidification and carbonate chemistry in the Great Barrier Reef. The research aims to determine how exposure to ocean acidification is changing on the reefs and identify drivers of variability. Observations from sites along the Great Barrier Reef and models show that seasonal changes in carbonate chemistry are linked to a winter source of CO2 on the Great Barrier Reef shelf. Coastal inputs also influence carbonate chemistry. The research uses data from ship surveys, moorings and sampling to improve biogeochemical models of the region from individual reef to whole Great Barrier Reef scales.Ocean Acidification Summary for Policymakers (2013)

The Ocean Acidification Summary for Policymakers was published in 2013. It summarises key findings from the Ocean in a High CO2 World Symposium, which is sponsored by the International Geosphere-Biosphere Programme, the Intergovernmental Oceanographic Commission of UNESCO and the Scientific Committee on Oceanic Research.
For more information
http://ocean-acidification.net/
Summary for Policymakers
http://www.igbp.net/publications/summariesforpolicymakers/summariesforpolicymakers/oceanacidificationsummaryforpolicymakers2013.5.30566fc6142425d6c9111f4.html
Ocean acidification & its prevention

Ocean acidification is caused by carbon dioxide emissions dissolving in oceans and reacting to form carbonic acid, increasing hydrogen ions and acidity. This impacts marine organisms like depressing immune responses in mussels and causing coral bleaching. The biggest contributors are livestock farming and fossil fuel use, so reducing meat consumption, using less energy, driving less, and choosing renewable energy can help prevent further acidification. Making sustainable consumer choices regarding plastics, water usage, and manufactured goods can also reduce the carbon footprint driving ocean acidification.
Ocean Acidification

This is a small presentation on ocean acidification.It is a compilation of all materials(including present information) I collected related to it, any new information beside this or concerning it please comment.
Ocean acidification __methane

Conclusion
While methane may be broken down into a less harmful form, when methane is oxidized the problem doesn't simply go away. Methane oxidizes into carbon dioxide both in seawater and in the atmosphere, adding to the carbon dioxide levels in both these realms. As we have seen, carbon dioxide in seawater contributes to ocean acidity, while atmospheric carbon dioxide traps heat and promotes warming – though not quite to the extent that methane would – and as atmospheric CO2 levels rise, so too will the CO2 that is absorbed into the ocean. It is a classic feedback loop that feeds into itself, having the potential to accelerate global warming and climate change to a tipping point – the point of no return.
Ocean Acidification Expert Forum Program

This document provides information about an Ocean Acidification Expert Forum held in February 2015 at the Victoria Conference Centre. It includes the agenda, speaker biographies, and context about ocean acidification. The two-day forum brought together international experts to review research on ocean acidification, identify key research needs for Canada, and establish a way forward for a coordinated Canadian research effort. The goal was to produce a white paper to guide research across sectors within Canada and internationally.
Paper discussion Fabry et al. 2008

1) Increased atmospheric CO2 is being absorbed by the oceans, raising CO2 levels and lowering pH, a process known as ocean acidification.
2) This affects calcifying organisms by decreasing carbonate ion concentrations needed to form calcium carbonate shells and skeletons. It impacts organisms' ability to calcify and causes dissolution of shells.
3) Pteropods, an important food source for salmon and other species, are especially vulnerable as they secrete the more soluble form of calcium carbonate, aragonite. Ocean acidification is restricting their habitat distribution.
Hypoxia and Ocean Acidification project

Dr. Francis Chan's 2012-2014 Oregon Sea Grant-supported project, "Understanding, Forecasting and Communicating the Linkages Between Hypoxia and Ocean Acidification in Oregon's Coastal Ocean"
Ocean acidification

The ocean covers two thirds of the planet and provides half of the oxygen and 20% of the protein for the world's population. However, ocean pollution from human activities is causing problems like ocean acidification. Ocean acidification occurs as the ocean absorbs 25% of the carbon dioxide emitted, which causes the pH of the ocean to decrease as carbon dioxide dissolves in seawater. This chemical change affects over 25% of marine species by decreasing the carbonate ions that many shellfish and corals use to build their shells, putting them at risk of dissolving. To slow ocean acidification and its impacts, carbon dioxide emissions must be reduced.
Climate change

Climate change is caused by human activity that releases heat-trapping greenhouse gases like carbon dioxide and methane. Greenhouse gas levels are the highest they've been in 800,000 years and global temperatures are rising twice as fast as 50 years ago. Effects of climate change include rising temperatures, sea levels and droughts/heat waves that can worsen wildfires. Reducing fossil fuel use and increasing renewable energy and sustainability can help mitigate and adapt to climate change to reduce health impacts.
What's hot (20)
Richard Feeley presentation on ocean acidification

Richard Feeley presentation on ocean acidification
Ocean acidification will-the_reef_survive-non-narrated[1]![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Ocean acidification will-the_reef_survive-non-narrated[1]
C5.01: Ocean acidification and seawater carbon chemistry of the Great Barrier...

C5.01: Ocean acidification and seawater carbon chemistry of the Great Barrier...
Ocean Acidification Summary for Policymakers (2013)

Ocean Acidification Summary for Policymakers (2013)
Similar to Scott Doney's Ocean Acidification presentation, April 2013 Hourglass Brasserie
Discussant: Corinne Le Quéré, Director, Tyndall Centre for Climate Change Res...

The document discusses several emerging issues impacting open oceans from a science perspective, including:
1) Surface salinity and temperature trends from 1950-2011 show changes due to climate change.
2) Ocean acidification has increased since 1979-2007 due to rising atmospheric CO2 levels.
3) Oxygen levels in oceans' top 1000 meters have declined between the 1990s and 1970s.
Peter Ridd

The document discusses potential threats to the Great Barrier Reef, including increased sediment and nutrient levels from agriculture as well as impacts from global warming. While these threats have concerned some, the author argues that the evidence shows the reef is in good condition overall and that the threats have been exaggerated. The author also expresses concerns about bias and lack of open debate in the scientific process around the reef's conservation.
International journal of applied sciences and innovation vol 2015 - no 1 - ...

Seagrasses act as important carbon sinks, trapping carbon for thousands of years and reducing the impacts of climate change and ocean acidification. They photosynthesize, removing carbon dioxide from water and oceans. However, seagrass meadows are declining globally at a rate four times faster than rainforests. While terrestrial forests receive more attention as carbon sinks, seagrasses also store large amounts of "blue carbon" and provide valuable ecosystem services. There is a need to increase awareness of seagrasses and protect these threatened coastal habitats.
OCEAN ACIDIFICATION soil science presentation.pptx

Ocean acidification is caused by carbon dioxide emissions from burning fossil fuels dissolving into the ocean and changing its chemistry. This leads to a decrease in ocean pH and increase in acidity. Impacts include loss of marine diversity and food sources as species that require calcium carbonate to grow, like corals, pteropods, and foraminifera, struggle under more acidic conditions. A case study from the Arctic Ocean found that pH levels have dropped 0.1 units over the past 30 years and are projected to decrease another 0.3 units by 2100 due to continued carbon dioxide emissions warming the oceans and allowing more absorption. Addressing ocean acidification will require reducing carbon dioxide emissions and improving ocean health through marine protected areas and sustainable fishing
Ocean Acification-Poster(ZSL)-2008

Ocean temperatures are rising, causing increased coral bleaching events, disease, and mortality. If greenhouse gas emissions are not reduced, ocean temperatures are projected to increase by 1.8-4°C by 2100, exceeding the tolerance of many coral species. Increasing temperatures, ocean acidification, sea level rise, and extreme weather events from climate change threaten widespread damage and decline of coral reef ecosystems. Immediate reductions in greenhouse gas emissions, especially CO2, are needed to avoid pushing coral reefs past critical environmental tipping points that could lead to ecosystem collapse.
UTSpeaks: Keeping Seas Green

Can preserving humble seagrass help protect us from the extremes of human-induced climate change?
The oceans have long been recognised by science as vital for capturing carbon and renewing the atmospheric balance that preserves life on earth. While vast amounts carbon are captured by phytoplankton, less well known has been the role played by seagrasses in storing carbon, cleansing the air and providing essential habitat for marine life.
Based on latest UTS marine research, this public lecture reveals the essential place of seagrasses in global ecology, the growing threats to its continued viability and the work that is being done to rehabilitate the areas of seagrass habitat already lost.
Professor Bill Gladstone
Marine biologist Bill Gladstone applies scientific understanding to solve problems in marine conservation and environmental management. His interests lie in assessing conservation values in marine ecosystems, the selection and management of marine parks, and community participation in marine conservation. He has worked throughout NSW, the Great Barrier Reef, Torres Strait, the Coral Triangle, and the Middle East.
Dr Peter Macreadie
Marine ecologist Peter Macreadie is a UTS Chancellor’s Postdoctoral Research Fellow. His research cover a wide range of systems; from deep-sea reefs to intertidal oyster reefs. Peter’s current research focuses on seagrasses to better understand how their resilience to climate change can be improved, and how can we capitalise on their ability to capture and store atmospheric carbon.
Professor Peter Ralph
Peter Ralph has been working with seagrasses since the early 90’s, when he pioneered the use of optical methods of measuring photosynthesis to examine the impact of pollution on seagrass health. More recently, he is developing new tools to assess the ability of an entire seagrass meadow to fix carbon. This work is now part of an international research agenda lead by the International Union for Conservation of Nature (IUCN) to demonstrate the importance of seagrasses in the global carbon cycle.
UTSpeaks is an annual free public lecture series presented by UTS experts discussing a range of important issues confronting contemporary Australia.
Use the hashtag #utspeaks to tweet about the lecture on Twitter.
Intro sustainable energy

This document provides an overview of a course on sustainable energy that aims to equip teachers to discuss complex issues around energy and climate change. The course covers motivations for changing energy production, including finite fossil fuel resources, energy security concerns, and the environmental impacts of climate change. It outlines sessions that will discuss energy consumption trends, various renewable and non-renewable technology options, and developing balanced future energy plans.
Coral triangle vs climate change

The document discusses the threats that climate change poses to coral reef ecosystems in the Coral Triangle region. It notes that rising carbon dioxide levels and global warming are causing ocean acidification and increased thermal stress that can lead to mass coral bleaching events. When sea temperatures rise just 1°C above normal for an extended period, represented by a degree heating week measurement above 8, widespread coral bleaching and death is likely to occur. The 1998 bleaching event across the Western Indian Ocean saw 95% of corals die on some reefs. If carbon dioxide levels continue increasing rapidly, the future state of coral reefs will be severely threatened.
Caballero-Alfonso et al. 2015

The document discusses biogeochemical and environmental drivers of coastal hypoxia in the Baltic Sea. It analyzes long-term trends in dissolved oxygen levels and factors influencing hypoxia at 33 coastal sites over multiple decades. Nutrient levels, temperature, stratification, and chlorophyll-a varied significantly between sites and impacted bottom water oxygen concentrations. While some sites showed improving oxygen trends following nutrient reductions, most sites continued worsening due to eutrophication despite remediation efforts. Managing nutrient inputs was identified as key to enabling oxygen recovery in coastal ecosystems.
KVA Symposium ocean acidification Cornell 2014

This document discusses what is known about the costs of ocean acidification for future Swedish society. It notes that while little is known, enough is understood to inform policy choices. Ocean acidification is part of complex global human-caused changes and is essentially irreversible. Delaying climate change mitigation will cost more in the future than taking action now. Further research is needed on local effects within global frameworks to support precautionary and participatory policies.
Clad oct09 jdawson

This document provides an overview of competing theories for increases in freshwater dissolved organic carbon (DOC) concentrations. It summarizes long-term DOC data from a site in Scotland that shows increasing concentrations over time. It also discusses seasonal and spatial variability in DOC levels across the UK, Europe and North America. Finally, it outlines the main hypothesized drivers for rising DOC, including increased temperatures, changing hydrology, land use changes, and others, but notes the challenges in isolating single causes given the complex interactions between multiple potential factors.
AFS Position Paper and Policy on Mining and Fossil Fuel Extraction

This document is an American Fisheries Society position paper and policy on mining and fossil fuel extraction approved in 2015. It discusses the potential impacts of these industries on aquatic ecosystems, including water contamination, habitat alterations, and effects on fish and other aquatic organisms. These impacts can occur through mining activities like mountaintop removal, acid mine drainage, and tailings pond failures. Fossil fuel extraction and use also threaten water quality and contribute to climate change. The policy calls for more rigorous environmental assessments, public involvement, monitoring, and regulations to help ensure environmentally responsible development and protection of aquatic resources.
7 peabody s4 oa presentation2 - peabody 10-27-10

This document summarizes a presentation on ocean acidification and its potential effects on shellfish in Puget Sound. It discusses how increased carbon dioxide in the atmosphere is causing the pH of oceans to decrease through ocean acidification. This poses risks to shellfish and other calcium-dependent marine species as the acidity makes it harder for them to build shells. The document outlines monitoring projects in Puget Sound to study spatfall and planktonic larvae in relation to changing water chemistry from ocean acidification. While no impacts have been directly observed yet in Puget Sound shellfish populations, continued monitoring is important given the risks and potential effects on the local shellfish industry and ecosystem.
Sea level CO2

The document discusses the consequences of increasing carbon dioxide (CO2) levels and rising sea levels. It notes that global temperatures have risen 0.6°C over the past 150 years and are predicted to increase 1.5-5.8°C by 2100, and that sea levels have risen by about 120 meters in the last 20,000 years and 1-2 mm per year in the last century, with predictions of a 0.13-0.94 meter rise by 2100. Higher CO2 levels will also likely lead to changes in precipitation patterns and more intense rainfall, and have consequences for marine processes and organisms.
High Def

- Hood Canal is a long, L-shaped body of water located in Washington state that has experienced declining dissolved oxygen levels, particularly in southern Hood Canal.
- Multiple factors contribute to low dissolved oxygen in Hood Canal, including strong stratification that prevents mixing of deep waters, slow circulation, and high productivity that leads to decomposition of organic matter and oxygen depletion.
- Increasing human development in the area has elevated nutrient levels in Hood Canal waters, fueling more rapid algal growth and higher oxygen demand when this material decomposes.
North Pacific Gyre & Ocean Pollution

This is a presentation about the North Pacific Gyre & Ocean Pollution. There are 5 gyres and each are a collection of litter from all over the world. A gyre is a wind driven surface current.
final dissertation report

- The document discusses how giant icebergs can influence marine productivity in the Southern Ocean by providing an important source of iron-rich terrigenous material to surrounding waters. Field studies have shown that iceberg-delivered iron leads to increased primary production and a more developed food chain ecosystem.
- Remote sensing of chlorophyll concentrations revealed a 92% likelihood of higher chlorophyll values, up to 3 times background levels, within 30 days and up to 860km from giant icebergs. This suggests icebergs fertilize large areas of ocean waters.
- The melting and dispersion of iron-rich debris from icebergs supports a rich pelagic ecosystem, with higher concentrations of phytoplankton,
Miriam Kastner: Her findings on METHANE HYDRATES in Ocean Acidification Summ...

Atmospheric carbon dioxide (CO2) levels are rising as a result of human activities, such as fossil fuel burning, and are increasing the acidity of seawater. This process is known as ocean acidi cation. Historically, the ocean has absorbed approximately 30% of all CO2 released into the atmosphere
by humans since the start of the industrial revolution, resulting in a 26% increase in the acidity of the ocean1.
Ocean acidi cation causes ecosystems and marine biodiversity to change. It has the potential to affect food security and it limits the capacity of the ocean to absorb CO2 from human emissions. The economic impact of ocean acidi cation could be substantial.
Reducing CO2 emissions is the only way to minimise long-term, large-scale risks.
Climate, carbon and coral reefs

Tropical coral reefs cover an area of over 284 000 km2, providing habitat for thousands of species and yielding more than US$ 30 billion annually in global goods and services, such as coastline protection, tourism and food. Corals reefs are now threatened by the increasing concentrations of carbon dioxide in the atmosphere, while warmer sea temperatures are disturbing the delicate symbiosis between coral organisms and algae. For example, 16 per cent of all tropical coral reefs were killed off by thermal stress during a single extreme El Niño–Southern Oscillation event in 1997–1998. As a result of escalating atmospheric levels of carbon dioxide, more of this gas is being dissolved in the world’s oceans. This has already reduced ocean pH and the trend is projected to continue. Moreover, the altered ocean chemistry is expected to have major corrosive effects on marine ecosystems and to alter the calcification rates of corals, phytoplankton and other species.
L'acidification des océans / IGBP, IOC, SCOR

The document summarizes key findings from the Third Symposium on the Ocean in a High-CO2 World, which convened over 500 experts. The main points are:
1) Ocean acidification is increasing at an unprecedented rate due to human CO2 emissions and is affecting ecosystems and biodiversity. It has the potential to impact food security and limit the ocean's ability to absorb more CO2.
2) Research shows adverse effects on some organisms' ability to form shells and skeletons as well as reduced survival, growth, and reproduction. However, some organisms can tolerate or benefit from acidification.
3) If emissions continue high, large parts of the polar oceans will become corrosive by decades
Similar to Scott Doney's Ocean Acidification presentation, April 2013 Hourglass Brasserie (20)
Discussant: Corinne Le Quéré, Director, Tyndall Centre for Climate Change Res...

Discussant: Corinne Le Quéré, Director, Tyndall Centre for Climate Change Res...
International journal of applied sciences and innovation vol 2015 - no 1 - ...

International journal of applied sciences and innovation vol 2015 - no 1 - ...
OCEAN ACIDIFICATION soil science presentation.pptx

OCEAN ACIDIFICATION soil science presentation.pptx
AFS Position Paper and Policy on Mining and Fossil Fuel Extraction

AFS Position Paper and Policy on Mining and Fossil Fuel Extraction
Miriam Kastner: Her findings on METHANE HYDRATES in Ocean Acidification Summ...

Miriam Kastner: Her findings on METHANE HYDRATES in Ocean Acidification Summ...
Recently uploaded
Kinetic studies on malachite green dye adsorption from aqueous solutions by A...

Kinetic studies on malachite green dye adsorption from aqueous solutions by A...Open Access Research Paper
Water polluted by dyestuffs compounds is a global threat to health and the environment; accordingly, we prepared a green novel sorbent chemical and Physical system from an algae, chitosan and chitosan nanoparticle and impregnated with algae with chitosan nanocomposite for the sorption of Malachite green dye from water. The algae with chitosan nanocomposite by a simple method and used as a recyclable and effective adsorbent for the removal of malachite green dye from aqueous solutions. Algae, chitosan, chitosan nanoparticle and algae with chitosan nanocomposite were characterized using different physicochemical methods. The functional groups and chemical compounds found in algae, chitosan, chitosan algae, chitosan nanoparticle, and chitosan nanoparticle with algae were identified using FTIR, SEM, and TGADTA/DTG techniques. The optimal adsorption conditions, different dosages, pH and Temperature the amount of algae with chitosan nanocomposite were determined. At optimized conditions and the batch equilibrium studies more than 99% of the dye was removed. The adsorption process data matched well kinetics showed that the reaction order for dye varied with pseudo-first order and pseudo-second order. Furthermore, the maximum adsorption capacity of the algae with chitosan nanocomposite toward malachite green dye reached as high as 15.5mg/g, respectively. Finally, multiple times reusing of algae with chitosan nanocomposite and removing dye from a real wastewater has made it a promising and attractive option for further practical applications.
Promoting Multilateral Cooperation for Sustainable Peatland management

Presented by Agus Julianto on GLF Peatlands 2024 - 360 Degree Approach to Improving Capacity of Multiple Stakeholders to Manage Peatland Sustainably
Peatlands of Latin America and the Caribbean

Presented by The Global Peatlands Assessment: Mapping, Policy, and Action at GLF Peatlands 2024 - The Global Peatlands Assessment: Mapping, Policy, and Action
原版制作(Newcastle毕业证书)纽卡斯尔大学毕业证在读证明一模一样

学校原件一模一样【微信:741003700 】《(Newcastle毕业证书)纽卡斯尔大学毕业证》【微信:741003700 】学位证,留信认证(真实可查,永久存档)原件一模一样纸张工艺/offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原。
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
【主营项目】
一.毕业证【q微741003700】成绩单、使馆认证、教育部认证、雅思托福成绩单、学生卡等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
如果您处于以下几种情况:
◇在校期间,因各种原因未能顺利毕业……拿不到官方毕业证【q/微741003700】
◇面对父母的压力,希望尽快拿到;
◇不清楚认证流程以及材料该如何准备;
◇回国时间很长,忘记办理;
◇回国马上就要找工作,办给用人单位看;
◇企事业单位必须要求办理的
◇需要报考公务员、购买免税车、落转户口
◇申请留学生创业基金
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Evolving Lifecycles with High Resolution Site Characterization (HRSC) and 3-D...

The incorporation of a 3DCSM and completion of HRSC provided a tool for enhanced, data-driven, decisions to support a change in remediation closure strategies. Currently, an approved pilot study has been obtained to shut-down the remediation systems (ISCO, P&T) and conduct a hydraulic study under non-pumping conditions. A separate micro-biological bench scale treatability study was competed that yielded positive results for an emerging innovative technology. As a result, a field pilot study has commenced with results expected in nine-twelve months. With the results of the hydraulic study, field pilot studies and an updated risk assessment leading site monitoring optimization cost lifecycle savings upwards of $15MM towards an alternatively evolved best available technology remediation closure strategy.
Peatland Management in Indonesia, Science to Policy and Knowledge Education

Peatland Management in Indonesia, Science to Policy and Knowledge EducationGlobal Landscapes Forum (GLF)
Presented by Feri Johana at GLF Peatlands 2024 - 360 Degree Approach to Improving Capacity of Multiple Stakeholders to Manage Peatland SustainablyImproving the Management of Peatlands and the Capacities of Stakeholders in I...

Improving the Management of Peatlands and the Capacities of Stakeholders in I...Global Landscapes Forum (GLF)
Presented by Sonya Dewi at GLF Peatlands 2024 - 360 Degree Approach to Improving Capacity of Multiple Stakeholders to Manage Peatland SustainablyEnvironment Conservation Rules 2023 (ECR)-2023.pptx

Bangladesh Environment Conservation Rules 2023 (ECR-2023).
Optimizing Post Remediation Groundwater Performance with Enhanced Microbiolog...

Results of geophysics and pneumatic injection pilot tests during 2003 – 2007 yielded significant positive results for injection delivery design and contaminant mass treatment, resulting in permanent shut-down of an existing groundwater Pump & Treat system.
Accessible source areas were subsequently removed (2011) by soil excavation and treated with the placement of Emulsified Vegetable Oil EVO and zero-valent iron ZVI to accelerate treatment of impacted groundwater in overburden and weathered fractured bedrock. Post pilot test and post remediation groundwater monitoring has included analyses of CVOCs, organic fatty acids, dissolved gases and QuantArray® -Chlor to quantify key microorganisms (e.g., Dehalococcoides, Dehalobacter, etc.) and functional genes (e.g., vinyl chloride reductase, methane monooxygenase, etc.) to assess potential for reductive dechlorination and aerobic cometabolism of CVOCs.
In 2022, the first commercial application of MetaArray™ was performed at the site. MetaArray™ utilizes statistical analysis, such as principal component analysis and multivariate analysis to provide evidence that reductive dechlorination is active or even that it is slowing. This creates actionable data allowing users to save money by making important site management decisions earlier.
The results of the MetaArray™ analysis’ support vector machine (SVM) identified groundwater monitoring wells with a 80% confidence that were characterized as either Limited for Reductive Decholorination or had a High Reductive Reduction Dechlorination potential. The results of MetaArray™ will be used to further optimize the site’s post remediation monitoring program for monitored natural attenuation.
Overview of the Global Peatlands Assessment

Presented by Patrick Scheel at GLF Peatlands 2024 - The Global Peatlands Assessment: Mapping, Policy, and Action
Epcon is One of the World's leading Manufacturing Companies.

Epcon is One of the World's leading Manufacturing Companies. With over 4000 installations worldwide, EPCON has been pioneering new techniques since 1977 that have become industry standards now. Founded in 1977, Epcon has grown from a one-man operation to a global leader in developing and manufacturing innovative air pollution control technology and industrial heating equipment.
原版制作(Manitoba毕业证书)曼尼托巴大学毕业证学位证一模一样

学校原件一模一样【微信:741003700 】《(Manitoba毕业证书)曼尼托巴大学毕业证学位证》【微信:741003700 】学位证,留信认证(真实可查,永久存档)原件一模一样纸张工艺/offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原。
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
【主营项目】
一.毕业证【q微741003700】成绩单、使馆认证、教育部认证、雅思托福成绩单、学生卡等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
如果您处于以下几种情况:
◇在校期间,因各种原因未能顺利毕业……拿不到官方毕业证【q/微741003700】
◇面对父母的压力,希望尽快拿到;
◇不清楚认证流程以及材料该如何准备;
◇回国时间很长,忘记办理;
◇回国马上就要找工作,办给用人单位看;
◇企事业单位必须要求办理的
◇需要报考公务员、购买免税车、落转户口
◇申请留学生创业基金
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
RoHS stands for Restriction of Hazardous Substances, which is also known as t...

RoHS stands for Restriction of Hazardous Substances, which is also known as the Directive 2002/95/EC. It includes the restrictions for the use of certain hazardous substances in electrical and electronic equipment. RoHS is a WEEE (Waste of Electrical and Electronic Equipment).
Global Peatlands Map and Hotspot Explanation Atlas

Presented by Alexandra Barthelmes at GLF Peatlands 2024 - The Global Peatlands Assessment: Mapping, Policy, and Action
Lessons from operationalizing integrated landscape approaches

Presented by James Reed at "9th Landscape Sustainability Science Forum" on 11 May 2024
在线办理(lboro毕业证书)拉夫堡大学毕业证学历证书一模一样

学校原件一模一样【微信:741003700 】《(lboro毕业证书)拉夫堡大学毕业证学历证书》【微信:741003700 】学位证,留信认证(真实可查,永久存档)原件一模一样纸张工艺/offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原。
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
【主营项目】
一.毕业证【q微741003700】成绩单、使馆认证、教育部认证、雅思托福成绩单、学生卡等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
如果您处于以下几种情况:
◇在校期间,因各种原因未能顺利毕业……拿不到官方毕业证【q/微741003700】
◇面对父母的压力,希望尽快拿到;
◇不清楚认证流程以及材料该如何准备;
◇回国时间很长,忘记办理;
◇回国马上就要找工作,办给用人单位看;
◇企事业单位必须要求办理的
◇需要报考公务员、购买免税车、落转户口
◇申请留学生创业基金
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Microbial characterisation and identification, and potability of River Kuywa ...

Microbial characterisation and identification, and potability of River Kuywa ...Open Access Research Paper
Water contamination is one of the major causes of water borne diseases worldwide. In Kenya, approximately 43% of people lack access to potable water due to human contamination. River Kuywa water is currently experiencing contamination due to human activities. Its water is widely used for domestic, agricultural, industrial and recreational purposes. This study aimed at characterizing bacteria and fungi in river Kuywa water. Water samples were randomly collected from four sites of the river: site A (Matisi), site B (Ngwelo), site C (Nzoia water pump) and site D (Chalicha), during the dry season (January-March 2018) and wet season (April-July 2018) and were transported to Maseno University Microbiology and plant pathology laboratory for analysis. The characterization and identification of bacteria and fungi were carried out using standard microbiological techniques. Nine bacterial genera and three fungi were identified from Kuywa river water. Clostridium spp., Staphylococcus spp., Enterobacter spp., Streptococcus spp., E. coli, Klebsiella spp., Shigella spp., Proteus spp. and Salmonella spp. Fungi were Fusarium oxysporum, Aspergillus flavus complex and Penicillium species. Wet season recorded highest bacterial and fungal counts (6.61-7.66 and 3.83-6.75cfu/ml) respectively. The results indicated that the river Kuywa water is polluted and therefore unsafe for human consumption before treatment. It is therefore recommended that the communities to ensure that they boil water especially for drinking.
Enhanced action and stakeholder engagement for sustainable peatland management

Enhanced action and stakeholder engagement for sustainable peatland managementGlobal Landscapes Forum (GLF)
Presented by Jan Peters at GLF Peatlands 2024 - The Global Peatlands Assessment: Mapping, Policy, and ActionWildlife-AnIntroduction.pdf so that you know more about our environment

All about wildlife, if you are fond of animals and wildlife then do follow us for more content like this
Recently uploaded (20)
Kinetic studies on malachite green dye adsorption from aqueous solutions by A...

Kinetic studies on malachite green dye adsorption from aqueous solutions by A...
Promoting Multilateral Cooperation for Sustainable Peatland management

Promoting Multilateral Cooperation for Sustainable Peatland management
Evolving Lifecycles with High Resolution Site Characterization (HRSC) and 3-D...

Evolving Lifecycles with High Resolution Site Characterization (HRSC) and 3-D...
Peatland Management in Indonesia, Science to Policy and Knowledge Education

Peatland Management in Indonesia, Science to Policy and Knowledge Education
Improving the Management of Peatlands and the Capacities of Stakeholders in I...

Improving the Management of Peatlands and the Capacities of Stakeholders in I...
Environment Conservation Rules 2023 (ECR)-2023.pptx

Environment Conservation Rules 2023 (ECR)-2023.pptx
Optimizing Post Remediation Groundwater Performance with Enhanced Microbiolog...

Optimizing Post Remediation Groundwater Performance with Enhanced Microbiolog...
Epcon is One of the World's leading Manufacturing Companies.

Epcon is One of the World's leading Manufacturing Companies.
RoHS stands for Restriction of Hazardous Substances, which is also known as t...

RoHS stands for Restriction of Hazardous Substances, which is also known as t...
Global Peatlands Map and Hotspot Explanation Atlas

Global Peatlands Map and Hotspot Explanation Atlas
Lessons from operationalizing integrated landscape approaches

Lessons from operationalizing integrated landscape approaches
Microbial characterisation and identification, and potability of River Kuywa ...

Microbial characterisation and identification, and potability of River Kuywa ...
world-environment-day-2024-240601103559-14f4c0b4.pptx

world-environment-day-2024-240601103559-14f4c0b4.pptx
Enhanced action and stakeholder engagement for sustainable peatland management

Enhanced action and stakeholder engagement for sustainable peatland management
Wildlife-AnIntroduction.pdf so that you know more about our environment

Wildlife-AnIntroduction.pdf so that you know more about our environment
Scott Doney's Ocean Acidification presentation, April 2013 Hourglass Brasserie
- 1. Ocean Acidification Scott Doney Woods Hole Oceanographic Institution
- 2. Fate of Anthropogenic CO2 Emissions (2000-2010) LeQuere et al. Nature Geosciences 2009; Global Carbon Project 2011 ~10 billion tons carbon per year+ Atmosphere 47% Land 27% Oceans 26%
- 3. Marine Life Susceptible to Ocean Acidification -Reduced shell formation -Habitat loss -Less available prey
- 4. Negative Impacts of CO2 on Mollusks Anne Cohen & Dan McCorkle WHOI (2012) Present (Vineyard Sound) Future (estuaries) Eastern Oyster Larvae 10 microns
- 5. Collapse of Pacific NW Oyster Hatcheries
- 6. U.S. ex-vessel revenue ~$4 Billion/year NOAA NMFS; Cooley & Doney ERL 2009 Valuable Commercial Shellfish Fisheries
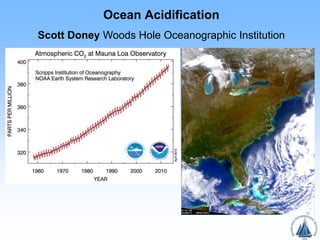
- 7. Acidification along U.S. East Coast Depth(m) Wang et al. Limnology & Oceanography 2013 Distance From Shore (km) acidic waters
- 8. Other Local Sources of Acidification Doney et al. PNAS 2007; Doney Science 2010; Kelly et al. Science 2011