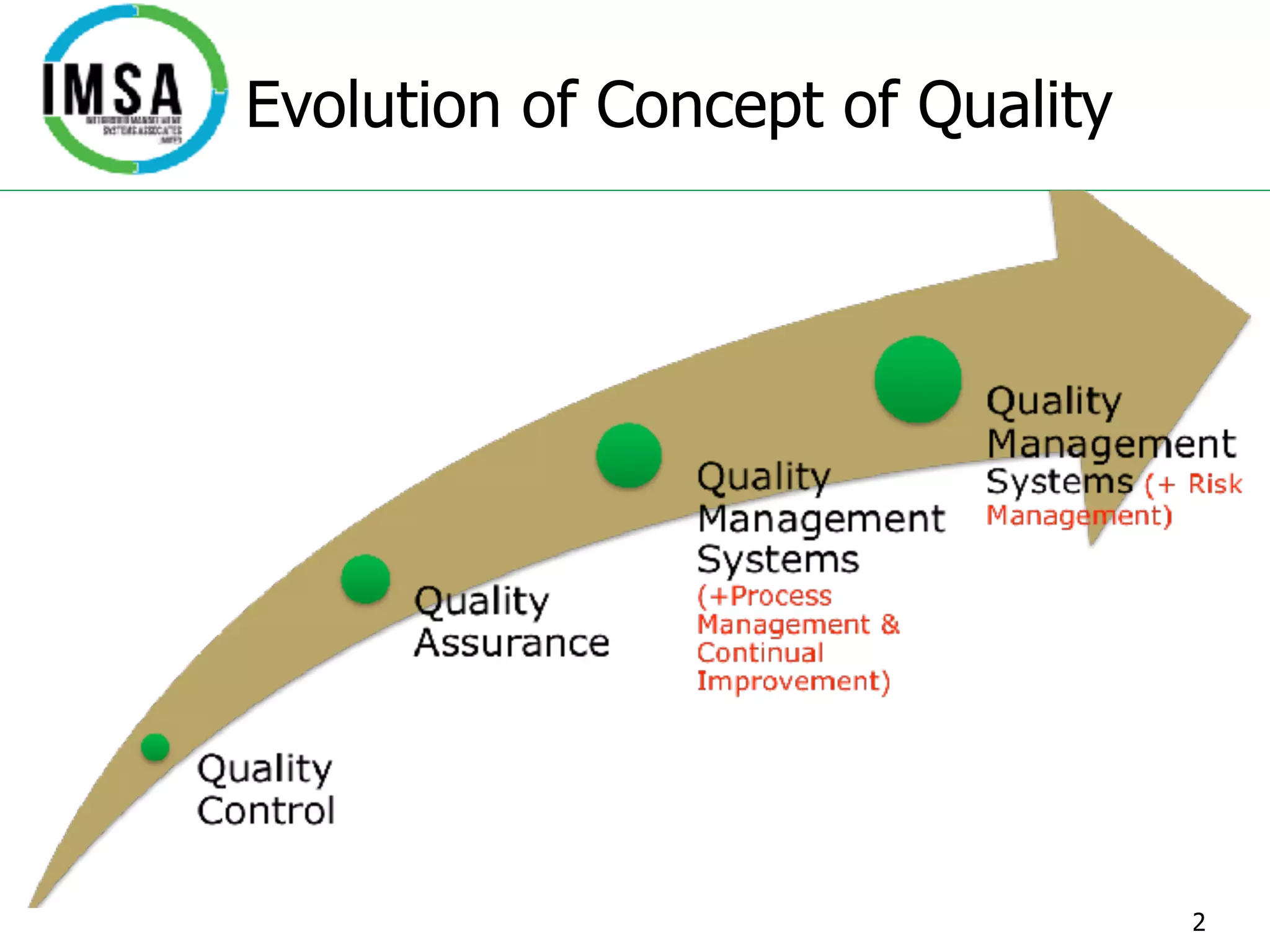
This document discusses quality management systems and standards in the aviation industry. It provides an overview of the evolution of quality management concepts and supporting standards such as ISO 9001. The key principles of quality management are outlined, including management leadership, risk management, process approach and continual improvement. The structure and requirements of the ISO 9001 quality management system standard are summarized. Industry-specific standards for aviation, space and defense like AS 9100 are also introduced.