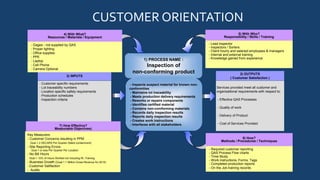
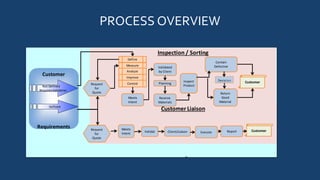
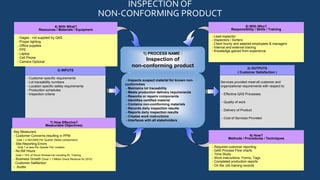
Quality Assurance Services, Inc. (QAS) provides quality control services including inspection, sorting, containment, and rework to meet ISO 9001:2008 standards. Its mission is to be a successful supplier committed to continuous improvement, customer satisfaction, and long-term partnerships. QAS inspects non-conforming products using customer requirements and production schedules, containing defects and reworking components as needed. Key metrics include customer concerns, reporting errors, and no bill hours to ensure effectiveness.