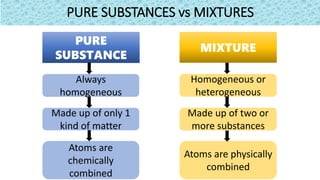
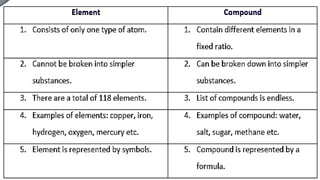
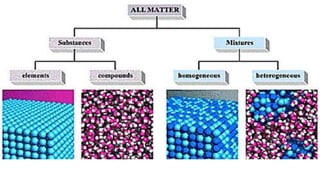
The document discusses the classification of matter. It defines matter as anything that occupies space and has mass. It provides examples of elements like sodium, chlorine, oxygen, and compounds like water, salt, sugar. Pure substances are classified as either elements or compounds. Elements are made of only one type of atom, while compounds are made of two or more elements chemically combined. The document seeks to distinguish between elements and compounds.