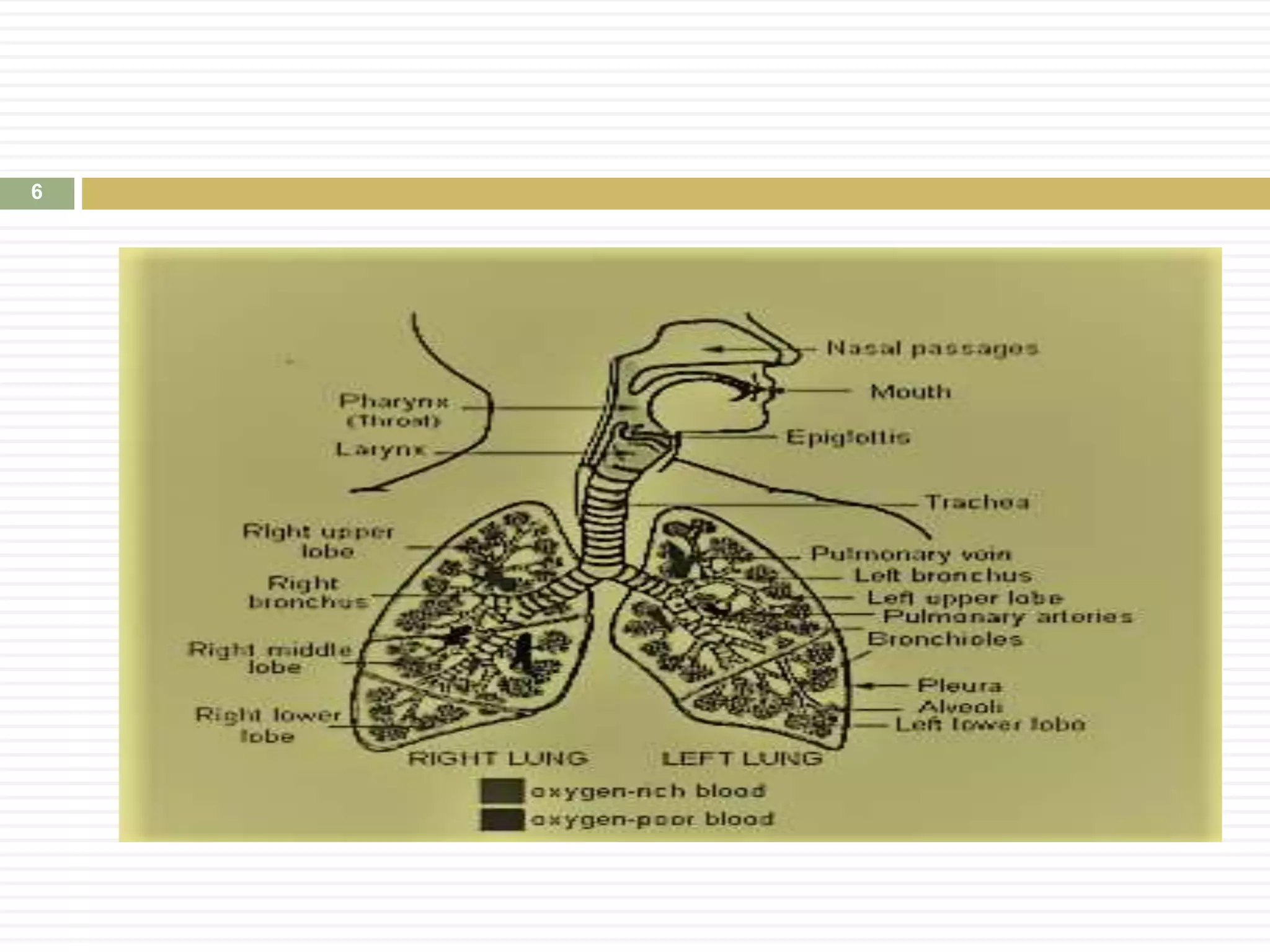
The document discusses pulmonary drug delivery systems focusing on their benefits, technologies, and challenges. Pulmonary delivery is advantageous in targeted treatment of respiratory diseases, offering rapid effects and minimizing systemic side effects, but faces issues like low efficiency and formulation stability. Various aerosol types, inhalation devices, and methods for evaluating drug delivery effectiveness are also reviewed.