Pre-shipment inspection report on July 27th,2016
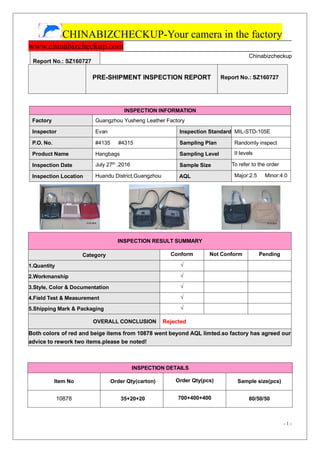
- 1. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 1 - Chinabizcheckup INSPECTION RESULT SUMMARY Category Conform Not Conform Pending 1.Quantity √ 2.Workmanship √ 3.Style, Color & Documentation √ 4.Field Test & Measurement √ 5.Shipping Mark & Packaging √ OVERALL CONCLUSION Rejected Both colors of red and beige items from 10878 went beyond AQL limted.so factory has agreed our advice to rework two items.please be noted! INSPECTION DETAILS Item No Order Qty(carton) Order Qty(pcs) Sample size(pcs) 10878 35+20+20 700+400+400 80/50/50 PRE-SHIPMENT INSPECTION REPORT Report No.: SZ160727 INSPECTION INFORMATION Factory Guangzhou Yusheng Leather Factory Inspector Evan Inspection Standard MIL-STD-105E P.O. No. #4135 #4315 Sampling Plan Randomly inspect Product Name Hangbags Sampling Level II levels Inspection Date July 27th ,2016 Sample Size To refer to the order Inspection Location Huandu District,Guangzhou AQL Major:2.5 Minor:4.0
- 2. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 2 - 18678 30+20 600+400 80/50 18679 33+20 500+300 50/50 Defects Description Item Defect description Critical Major Minor 10878 Zipper head is stuck 0 5 12 Open seam 0 6 10 Untrimmed thread residue 0 0 6 18678 Mixed color 0 1 1 Open seam 0 0 1 Untrimmed thread residue 0 0 4 18679 Poor sewing 0 0 1 Defect pictures from 10878 The zipper head is got stuck when it’s moved The zipper head is got stuck when it’s moved
- 3. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 3 - Open seam Open seam Open seam Open seam Open seam Open seam
- 4. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 4 - Open seam Open seam Untrimmed thread residue Untrimmed thread residue Defect pictures from 18678 Mixed color on the leather Mixed color on the leather
- 5. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 5 - Open seam Untrimmed thread residue Untrimmed thread residue Untrimmed thread residue Untrimmed thread residue Untrimmed thread residue Defect pictures from 18679
- 6. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 6 - Poor sewing Untrimmed thread residue Pack&Product Information for 10878 Finished goods Carton box view
- 7. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 7 - Front mark(black) Front mark (beige) Front mark(red) Side mark Side mark(black) Side mark(beige)
- 8. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 8 - Side mark(red) Opening carton box to view Fluting paper with seven layers Opening carton box to view Poly bag view Pack view
- 9. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 9 - Pack view Poly bag view Pack view Pack view Poly bag view Three color of the items view
- 10. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 10 - Left side of item is sample and right side of item is bulk production Left side of item is sample and right side of item is bulk production Three color of the items view Logo view Product view in details Product view in details
- 11. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 11 - Product view in details Product view in details Hangtag view Hangtag view Bar code scanned Bar code scanned
- 12. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 12 - Hangtag view Hangtag view Bar code scanned Bar code scanned Hangtag view Hangtag view
- 13. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 13 - Bar code scanned Bar code scanned Three colors of the items view Product view in details Product view in details Product view in details
- 14. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 14 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 15. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 15 - Product view in details Product view in details Product view in details Product view in details Product view in details Fabric label with bar code
- 16. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 16 - Bar code scanned Fabric label with bar code Bar code scanned Fabric label with bar code Bar code scanned Non-woven bag view
- 17. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 17 - Logo view Non-woven bag view Product view Product view Nil Product view Nil
- 18. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 18 - Measurement Date from 10878 Gross weight check Gross weight show Gross weight check Gross weight show
- 19. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 19 - Gross weight check Gross weight show Size measurement Size measurement Size measurement Size measurement
- 20. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 20 - Size measurement Size measurement Size measurement Size measurement Size measurement Size measurement
- 21. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 21 - Size measurement Size measurement Size measurement Size measurement
- 22. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 22 - Size measurement Single net weight show Single net weight show Single net weight show Pack and Product Information from 18678 Finished goods Finished goods
- 23. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 23 - Carton box view Front mark(camel) Side mark (camel) Front mark(black) Front mark(black) Side mark(black)
- 24. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 24 - Pack view Fluting paper with seven layers Pack view Poly bag view Poly bag view Two items views
- 25. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 25 - The approved color sample is matched with the bulk production The approved color sample is matched with the bulk production Products view in details Product view in details Product view in details Product view in details
- 26. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 26 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 27. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 27 - Product view in details Product view in details Product view in details Product view in details Product view in details Products view in details
- 28. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 28 - Product view in details Product view in details Product view in details Product view in details Product view in details Hangtag view
- 29. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 29 - Hangtag view Bar code scanned Bar code scanned Hangtag view Hangtag view Bar code scanned
- 30. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 30 - Bar code scanned Product view in details Products view in details Products view in details Product view in details Product view in details
- 31. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 31 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 32. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 32 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 33. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 33 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 34. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 34 - Product view in details Product view in details Product view in details Product view in details Product view in details Fabric label view
- 35. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 35 - Product view in details Products view in details Product view Product view Measurement Date from 18678
- 36. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 36 - Size measurement Size measurement Size measurement Size measurement Size measurement Size measurement
- 37. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 37 - Gross weight check Gross weight show Gross weight check Gross weight show Size measurement Size measurement
- 38. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 38 - Size measurement Size measurement Size measurement Size measurement Size measurement Size measurement
- 39. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 39 - Size measurement Size measurement Size measurement Size measurement Single net weight show Single net weight show
- 40. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 40 - Pack and Product Information from 18679 Finished goods Finished goods Sid mark(black) Side mark(black)
- 41. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 41 - Front mark(camel) Side mark(camel) Pack view Fluting paper with seven layers Pack view Pack view
- 42. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 42 - Pack view Two items view Poly bag view Poly bag view Products view in details Product view in details
- 43. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 43 - Product view in details Hangtag view Hangtag view Bar code scanned Bar code scanned Product view in details
- 44. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 44 - Product view in details Hangtag view Hangtag view Bar code scanned Bar code scanned Products view in details
- 45. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 45 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 46. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 46 - Products view in details Products view in details Products view in details Products view in details Products view in details Product view in details
- 47. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 47 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 48. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 48 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 49. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 49 - Product view in details Fabric label view Product view in details Product view in details Product view in details Product view in details
- 50. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 50 - Products view in details Product view in details Products view in details Product view in details Product view in details Product view in details
- 51. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 51 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 52. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 52 - Product view in details Product view in details Product view in details Product view in details Product view in details Product view in details
- 53. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 53 - Product view in details Products view in details Product view in details Products view in details Products view in details Non-woven bag view
- 54. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 54 - Non-woven bag view Product view Product view Product view Product view Product view
- 55. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 55 - Measurement Date from 18679 Size measurement Size measurement Size measurement Size measurement Size measurement Size measurement
- 56. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 56 - Gross weight check Gross weight show Gross weight check Gross weight show Single net weigh show Single net weight show
- 57. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 57 - Size measurement Size measurement Size measurement Size measurement Size measurement Size measurement
- 58. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 58 - Size measurement Size measurement Size measurement Size measurement Size measurement Size measurement
- 59. CHINABIZCHECKUP-Your camera in the factory www.chinabizcheckup.com Report No.: SZ160727 - 59 - Nil Size measurement Nil ------------------------------------------------------------------------End of Report-----------------------------------------------------------------------