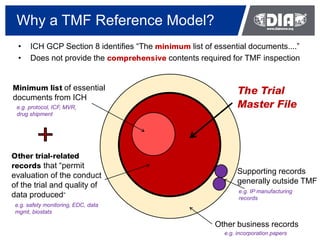
The webinar provided an overview of applying the TMF Reference Model in organizations. It discussed what the TMF Reference Model is, how to implement it in practical steps, and specific content considerations. Attendees were polled on their familiarity with and use of the TMF Reference Model. The presentation covered surveying current processes, differentiating issues, preparing tools like the reference model, and working through implementation including maintaining the new standard.