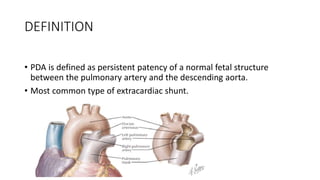
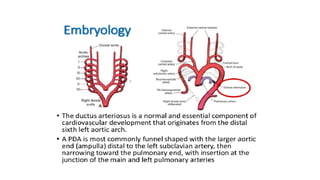
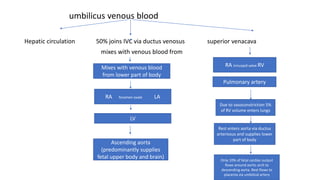
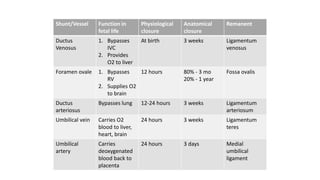
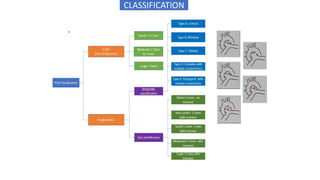
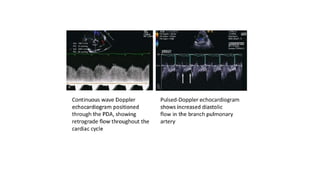
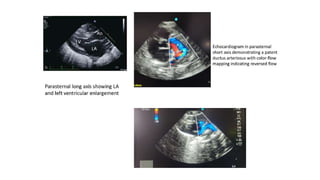
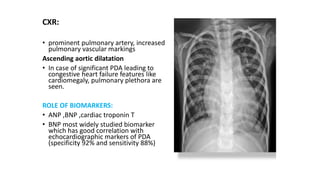
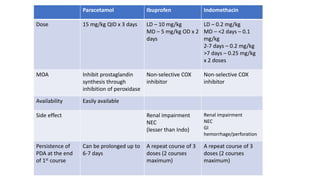
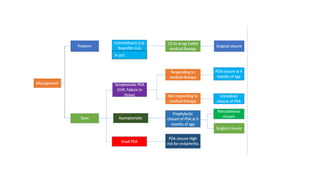
The document discusses patent ductus arteriosus (PDA) in preterm infants. It defines PDA and describes the pathophysiology in preterm infants as delayed closure due to low intrinsic ductal tone and immature tissues. Clinical signs include bounding pulses, murmurs, and respiratory issues. Echocardiogram confirms the diagnosis. Management involves initial conservative measures, then drugs like indomethacin that inhibit prostaglandin and close the ductus. If medical therapy fails, surgical ligation is recommended. Complications of a large PDA include pulmonary overcirculation, systemic hypotension, and increased risks of other morbidities.