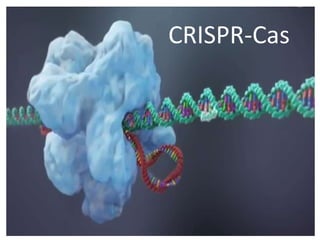
Multi Target Gene Editing using CRISPR Technology for Crop Improvement
- 1. CRISPR-Cas
- 2. Plant Biotechnology Centre , DBSKKV, Dapoli A Presentation On Multi Target Gene Editing using CRISPR Technology for Crop Improvement Presented by Gajare Tushar .P. Reg.No. 0030 Sr.Msc. Agricultural Biotechnology Plant Biotechnology Centre College of Agriculture, Dapoli
- 3. CRISPR-Cas • Introduction • CRISPR • Cas • How it works? • CRISPR-Cas: A tool for Genetic Engineering • History • Applications • Applications in Crop Improvement • Case studies • Advantages • Disadvantages • Safety issues • Future apsects • Conclusion • References Plant Biotechnology Centre , DBSKKV, Dapoli
- 4. Introduction • Genome editing : Genome editing or gene editing, is a group of technologies that give scientists the ability to change an organism's DNA. • These technologies allow genetic material to be added, removed, or altered at particular locations in the genome. • Several approaches to genome editing have been developed. Eg: CRISPR, TALENs, ZFNs Plant Biotechnology Centre , DBSKKV, Dapoli
- 5. Introduction • What is CRISPR-Cas? • CRISPR-Cas(9) is a unique technology that enables us to edit parts of the genome by removing, adding or altering sections of the DNA sequences. • It is currently the simplest, most versatile and precise method of genetic manipulation and is therefore causing a buzz in the science world. Plant Biotechnology Centre , DBSKKV, Dapoli
- 6. CRISPR • Clustered Regularly Interspaced Short Palindromic Repeats. • It is a family of DNA sequences found within the genomes of prokaryotic organisms such as bacteria and archaea. • Derived from DNA fragments from viruses that have previously infected the prokaryote and are used to detect and destroy DNA from similar viruses during subsequent infections. • Plays a key role in the antiviral defence system of prokaryotes. Plant Biotechnology Centre , DBSKKV, Dapoli
- 7. Cas9 • CRISPR-associated protein 9. • An enzyme that uses CRISPR sequences as a guide to recognize and cleave specific strands of DNA that are complementary to the CRISPR sequence. • Cas9 enzymes together with CRISPR sequences form the basis of a technology known as CRISPR/Cas9 that can be used to edit genes within organisms. Plant Biotechnology Centre , DBSKKV, Dapoli Cas9 protein molecule
- 8. Plant Biotechnology Centre , DBSKKV, Dapoli
- 9. How it works? • In this system, the endonuclease of Cas9 is directed to DNA targets by a guideRNA . • The ribonucleoprotein complex of Cas9 and gRNA recognizes the DNA sequence that is complementary to the 5’-end of the guideRNA. • The DNA sequence is then cleaved. • Thus disintegrating the viral DNA. Plant Biotechnology Centre , DBSKKV, Dapoli
- 10. Plant Biotechnology Centre , DBSKKV, Dapoli
- 11. CRISPR-Cas(9): As a Tool • Cas9 (acts as a pair of ‘molecular scissors’ ) cuts the two strands of DNA at a specific location in the genome. • The bits of DNA are then be added or removed. • gRNA consists of a small piece of predesigned RNA sequence (about 20 bases long) located within a longer RNA scaffold. • Pre-designed sequence ‘guides’ Cas9 to the right part of the genome. • This makes sure that the Cas9 enzyme cuts at the right point in the genome. • The guide RNA is designed to find and bind to a specific sequence in the DNA. • gRNA has bases complementary to the target DNA molecule. • the guide RNA will only bind to the target sequence and no other regions of the genome. • The scaffold part binds to DNA . • The Cas9 follows the guide RNA to the same location in the DNA sequence and makes a cut across both strands of the DNA. • At this stage the cell recognises that the DNA is damaged and tries to repair it. • DNA repair machinery is used to introduce changes to one or more genes the genome by adding , deleting or replacing the genes. Plant Biotechnology Centre , DBSKKV, Dapoli
- 12. Plant Biotechnology Centre , DBSKKV, Dapoli Cas9 (acts as a pair of ‘molecular scissors’ ) cuts the two strands of DNA at a specific location in the genome. The bits of DNA are then be added or removed gRNA It ‘guides’ Cas9 to the right part of the genome. This makes sure that the Cas9 enzyme cuts at the right point in the genome. The scaffold part binds to DNA . The Cas9 follows the guide RNA to the same location in the DNA sequence and makes a cut across both strands of the DNA. cell recognises that the DNA is damaged and tries to repair it. DNA repair machinery is used Genome is edited by adding , deleting or replacing the genes.
- 13. Plant Biotechnology Centre , DBSKKV, Dapoli
- 14. History • The discovery of clustered DNA repeats occurred independently in three parts of the world. Plant Biotechnology Centre , DBSKKV, Dapoli 1987 • Yoshizumi Ishino et al. • Accidentally cloned part of a CRISPR together with the iap gene, the target of interest. • They studied the relation of "iap“ to the bacterium E. coli. 1993 • Mycobacterium tuberculosis • In Neterlands two research articles • Diversity of cluster of interrupted direct repeats in different strains • spoligotyping 1993 • Francisco Mojica • Observed and studied the function of repeats in Haloferax and Haloarcula species
- 15. 2000 •Mojica •Survey of scientific literature 2001 •Mojica and Ruud Jansen •Proposed the acronym CRISPR 2002 •Tang, et al. •showed evidence that CRISPR repeat regions from the genome of Archaeoglobus fulgidus were transcribed into long RNA molecules that were subsequently processed into unit-length small RNAs, plus some longer forms of 2,3, or more spacer-repeat units. Plant Biotechnology Centre , DBSKKV, Dapoli
- 16. Plant Biotechnology Centre , DBSKKV, Dapoli 2013 •Cong et al •First event of CRISPR/Cas 9 in Eukaryotes •Mao et al •designed two sgRNAs for the photosynthesis in Arabidopsis thaliana. 2014 •Wang et al •Cas 9 used for functional screening 2015 •Ousterout et al •Multiplex CRISPR/Cas 9
- 17. Plant Biotechnology Centre , DBSKKV, Dapoli
- 18. Applications • Biomedical research • Treating diseases and genetic disorders • Crop Improvement Plant Biotechnology Centre , DBSKKV, Dapoli
- 19. Applications in Crop Improvement • Gene knockouts: • Stress resistance: • Inducing crop diversity: • In nitrogen fixation: • To reduce apple acidity: • Inducing polyploidy: • For suppressing viral infection: • Muti target gene editing/ Multiplex Genome editing : Plant Biotechnology Centre , DBSKKV, Dapoli
- 20. • Gene knockouts: To eliminate undesirable genes • Stress resistance: Multiplexing ability to impart biotic and abiotic stress tolerance. Eg: virus-induced gene silencing (VIGS) in tomato. • Inducing crop diversity: • To create a high degree of genetic variability at a precise locus in the genome of the crop plants. • It is a potential tool for multiplexed reverse and forward genetic study. • Genome editing allows precise and predictable modifications directly in elite cultivars or accessions, saving the time consuming backcrossing procedure in conventional breeding schemes. Plant Biotechnology Centre , DBSKKV, Dapoli
- 21. • In nitrogen fixation: • To reduce apple acidity: • Inducing polyploidy: • For the creation and use of novel allelic variants for breeding in crops. • Production of haploids. • Generating Polyploidy • E.g: Potato and wheat. • For suppressing viral infection: Highly conserved sequences of Gemini viruses have been targeted using CRISPR/Cas9 to good effect (Ali et al., 2016). Plant Biotechnology Centre , DBSKKV, Dapoli
- 22. Muti target gene editing/ Multiplex Genome editing : WHY ? Plant Biotechnology Centre , DBSKKV, Dapoli • In plants, cellular processes are fine-tuned by several genes. • Sometimes, mutating a single gene may not confer a desired phenotype because of the compensation effect produced by other genes in same gene family. • Hence, an upgraded editing system with improved efficiency is needed for multiplex gene editing in plants.
- 23. Plant Biotechnology Centre , DBSKKV, Dapoli
- 24. Case Studies Plant Biotechnology Centre , DBSKKV, Dapoli 1 Targeted Mutagenesis, Precise Gene Editing, and Site-Specific Gene Insertion in Maize Using Cas9 and Guide RNA. Svitashev, S., Young, J. K., Schwartz, C., Gao, H., Falco, S. C., & Cigan, A. M. (2015).
- 25. Plant Biotechnology Centre , DBSKKV, Dapoli • Crop : Zea mays • Target genes : Liguleless1 (LIG1) gene, male fertility genes (Ms26 and Ms45), and acetolactate synthase (ALS) genes (ALS1 and ALS2) • Cas9 promoter: ZmUbi • Codon optimazition of Cas9 : Maize • sgRNA promoter : ZmU6 • Transformation method : Biolistic transformation • Multiplex strategy : Co-delivery • Mutant efficiancy : 77–100% • Type of mutant : biallelic,heterozygous
- 26. Method Plant Biotechnology Centre , DBSKKV, Dapoli • Targeted mutagenesis, editing of endogenous maize genes, and site-specific insertion of a trait gene using Cas9-guide RNA technology is reported in Zea mays. • DNA vectors expressing maize codon-optimized Streptococcus pyogenes Cas9 endonuclease and single guide RNAs were co-introduced with or without DNA repair templates into maize immature embryos by biolistic transformation targeting five different genomic regions: upstream of the liguleless-1 gene (LIG), male fertility genes (MS26 and MS45) and 49 acetolactate synthase genes (ALS1 and ALS2).
- 27. Observations Plant Biotechnology Centre , DBSKKV, Dapoli • Mutations were subsequently identified at all sites targeted • Plants containing biallelic multiplex mutations at LIG, MS26 and MS45 were recovered.
- 28. Results Plant Biotechnology Centre , DBSKKV, Dapoli • Biolistic delivery of guide RNAs (as RNA molecules) directly into immature embryo cells containing pre-integrated Cas9 also resulted in targeted mutations. • Editing the ALS2 gene using either single-stranded oligonucleotides or double-stranded DNA vectors as repair templates yielded chlorsulfuron resistant plants. • Double-strand breaks generated by RNA guided Cas9 endonuclease also stimulated insertion of a trait gene at a site near liguleless-1 by homology-directed repair. • Progeny demonstrated expected Mendelian segregation of mutations, edits, and targeted gene insertions.
- 29. Plant Biotechnology Centre , DBSKKV, Dapoli
- 30. Conclusion Plant Biotechnology Centre , DBSKKV, Dapoli • The examples reported in this study demonstrate the utility of Cas9-guide RNA technology as a plant genome editing tool to enhance plant breeding and crop research needed to meet growing agriculture demands of the future
- 31. Case Study 2 Plant Biotechnology Centre , DBSKKV, Dapoli Lycopene Is Enriched in Tomato Fruit by CRISPR/Cas9-Mediated Multiplex Genome Editing Xindi Li 1 , Yanning Wang 1 , Sha Chen 2 , Huiqin Tian 1 , Daqi Fu 1 , Benzhong Zhu 1 , Yunbo Luo 1 and Hongliang Zhu 1 * (2018)
- 32. Plant Biotechnology Centre , DBSKKV, Dapoli • Crop : Tomato (Lycopersicon esculentum) • Target genes : 5 1. SGR1 2. lycopene ε-cyclase (LCY-E) 3. beta-lycopene cyclase (Blc) 4. lycopene β-cyclase 1(LCY-B1) 5. LCY-B2 • Target sites : 6 • Promoters: pAtU3d, pAtU3b, pAtU6-1,pAtU6-29 • Transformation method : Agrobacterium tumefaciens-mediated transformation • Vector : pYLCRISPR/Cas9-Lycopene vector (binary plasmid ) • Multiplex strategy : bidirectional strategy • Type of mutant : biallelic,homozygous
- 33. Plant Biotechnology Centre , DBSKKV, Dapoli • Numerous studies have been focusing on breeding tomato plants with enhanced lycopene accumulation, considering its positive effects of fruits on the visual and functional properties. • In this study, a bidirectional strategy: promoting the biosynthesis of lycopene, while inhibiting the conversion from lycopene to β-and α- carotene was used. • The accumulation of lycopene was promoted by knocking down some genes associated with the carotenoid metabolic pathway. • Finally, five genes were selected to be edited in genome by CRISPR/Cas9 system using Agrobacterium tumefaciens-mediated transformation. • Findings indicated that CRISPR/Cas9 is a site-specific genome editing technology that allows highly efficient target mutagenesis in multiple genes of interest. • The lycopene content in tomato fruit subjected to genome editing was successfully increased to about 5.1-fold. • The homozygous mutations were stably transmitted to subsequent generations.
- 34. Plant Biotechnology Centre , DBSKKV, Dapoli FIGURE : Selection of target genes and designing of CRISPR/Cas9 binary expression cassette. (A) A map of the target genes in the carotenoid metabolic pathway. The green boxes represent the key substances in the metabolic pathway. The red and orange boxes show the two substances, lycopene and β-carotene, respectively. A solid arrow indicates a direct effect, and a dashed arrow indicates an indirect effect. The selected target genes are represented by purple boxes, and the red asterisks represent the sites at which the target genes act on the pathway. G3P, glyceraldehyde 3-phosphate; DXS, 1-deoxy-D-xylulose 5-phosphate synthase; GGPPS, geranylgeranyl pyrophosphate synthase; PDS, phytoene desaturase; ZISO, z-carotene isomerase
- 35. Plant Biotechnology Centre , DBSKKV, Dapoli (B) Five target genes were selected according to the synthesis and metabolism pathways of lycopene, and six target sites were designed. The target sequences are marked in red, and small rectangle frames indicate the PAM. Straight lines and boxes are the introns and exons of the target genes, respectively.
- 36. Plant Biotechnology Centre , DBSKKV, Dapoli (C) Structures of the pYLCRISPR/Cas9-Lycopene binary vectors. HPT(−H) encodes hygromycin B phosphotransferase. The six targets designed are represented by solid boxes in different colors, and the promoters used for each target are shown.
- 37. Methodology Plant Biotechnology Centre , DBSKKV, Dapoli Plant Material (Leaves ) Selection of sgRNA Target Sequence pYLCRISPR/Cas9- Lycopene Vector Construction Plant Transformation (Agrobacterium- mediated ) DNA Extraction and Mutation Detection Cas9 and Off-Target Analysis Carotenoid Extraction and RT- HPLC Analysis Carotenoid Extraction and HPLC- MS Analysis Transmission Electron Microscope Analysis
- 38. Result Plant Biotechnology Centre , DBSKKV, Dapoli
- 39. Conclusion Plant Biotechnology Centre , DBSKKV, Dapoli • The results suggest that CRISPR/Cas9 system can be used for significantly improving lycopene content in tomato fruit with advantages such as high efficiency, rare off- target mutations, and stable heredity.
- 40. Advantages • Faster • Cheaper • More accurate • Efficient • Specific • Less occurance of unwanted effects than other existing genome editing methods. • Ability of multiple targeting simultaneously. Plant Biotechnology Centre , DBSKKV, Dapoli
- 41. DisAdvantages Plant Biotechnology Centre , DBSKKV, Dapoli • Potential threat to Environment. • Risk of Permanent Modification. • Concerns have been raised that off-target effects (editing of genes besides the ones intended) may obscure the results of a CRISRP gene editing experiment (the observed phenotypic change may not be due to modifying the target gene, but some other gene). • Modifications to CRISRP have been made to minimize the possibility of off-target effects. • In addition, orthogonal CRISPR experiments are recommended to confirm the results of agene editing experiment.
- 42. Issues Plant Biotechnology Centre , DBSKKV, Dapoli • Safety • Ethical • Social • Political • Acceptance
- 43. Future apsects Plant Biotechnology Centre , DBSKKV, Dapoli • Gene editing • Transcription • Chromosome structure Determination
- 44. Conclusion Plant Biotechnology Centre , DBSKKV, Dapoli • CRISPR-Cas(9) enables us to edit genome by targeting multiple genes in a single approach with – • Great ease • High specificity • Higher speed and accuracy. • Minimal or no off-targets side effects. • It is currently the simplest, most versatile and precise method of genetic manipulation and is therefore causing a buzz in the science world.
- 45. References Plant Biotechnology Centre , DBSKKV, Dapoli • Devashish Rath , Amlinger. L. , Rath. A. , Lundgren.M. (2015)The CRISPR-Cas immune system: Biology, mechanisms and applications. Biochime(17):119-128. • Tsai S..Q, Zheng Z., Nguyen N.T., Liebers M., Topkar V.V., Thapar V. (2015) GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nat Biotechnol. 33 (2):187–197. • Svitashev, S., Young, J. K., Schwartz, C., Gao, H., Falco, S. C., & Cigan, A. M. (2015). Targeted Mutagenesis, Precise Gene Editing, and Site-Specific Gene Insertion in Maize Using Cas9 and Guide RNA. Plant Physiology, 169(2), 931–945. doi:10.1104/pp.15.00793 • Lowder, L. G., Zhang, D., Baltes, N. J., Paul, J. W., Tang, X., Zheng, X., … Qi, Y. (2015). A CRISPR/Cas9 Toolbox for Multiplexed Plant Genome Editing and Transcriptional Regulation. Plant Physiology, 169(2), 971–985. • Yang, Q., Zhang, C., Chan, M., Zhao, D., Chen, J., Wang, Q., … Liu, Q. (2016). Biofortification of rice with the essential amino acid lysine: molecular characterization, nutritional evaluation, and field performance. Journal of Experimental Botany, 67(14), 4285–4296. doi:10.1093/jxb/erw209 • Ali Z., Ali S., Tashkandi M., Zaidi S.S., Mahfouz M.M. (2016) CRISPR/Cas9-mediated immunity to geminiviruses: differential interference and evasion. Sci Rep. 6. • Doench J.G., Fusi N., Sullender M., Hegde M., Vaimberg E.W., Donovan K.F., SmithI.,Tothova Z., Wilen C. (2016) Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR-Cas9. Nat Biotechnol. 34:184–191. • Gayatonde, V., &Vennela, P. R. (2017). CRISPR-Cas; A potential technique for crop improvement. Biotech Express. 4(34):34-38. • Antre S. H., Mandlik R. R., Guru P. N.(2017) CRISPR/Cas9 : A Game Changer Tool in Crop Improvement. Current Advances and Challenges in Agricultural Biotechnology(pp.). PRES’s College of Agricultural Biotechnology, Loni, Ahmednagar.
- 46. • Georges, F., & Ray, H. (2017). Genome Editing of Crops: A Renewed Opportunity for Food Security. GM crops & food, 0. • Arora, L., & Narula, A. (2017). Gene Editing and Crop Improvement Using CRISPR- Cas9 System. Frontiers in Plant Science, 8. • Soda, N., Verma, L., & Giri, J. (2017). CRISPR-Cas9 based plant genome editing: Significance, opportunities and recent advances. Plant Physiology and Biochemistry. • Li, X., Wang, Y., Chen, S., Tian, H., Fu, D., Zhu, B., … Zhu, H. (2018). Lycopene Is Enriched in Tomato Fruit by CRISPR/Cas9-Mediated Multiplex Genome Editing. Frontiers in Plant Science, 9. • Leong, K.-Y. B., Chan, Y.-H., Abdullah, W. M. A. N. W., Lim, S.-H. E., & Lai, K.-S. (2018). The CRISPR/Cas9 System for Crop Improvement: Progress and Prospects. Next Generation Plant Breeding. • Jaganathan, D., Ramasamy, K., Sellamuthu, G., Jayabalan, S., & Venkataraman, G. (2018). CRISPR for Crop Improvement: An Update Review. Frontiers in Plant Science, 9. • Xu, J., Hua, K., & Lang, Z. (2019). Genome editing for horticultural crop improvement. Horticulture Research, 6(1). doi:10.1038/s41438-019-0196-5 • Razzaq, A., Saleem, F., Kanwal, M., Mustafa, G., Yousaf, S., Imran Arshad, H. M., … Joyia, F. A. (2019). Modern Trends in Plant Genome Editing: An Inclusive Review of the CRISPR/Cas9 Toolbox. International Journal of Molecular Sciences, 20(16), 4045. Plant Biotechnology Centre , DBSKKV, Dapoli
- 47. Plant Biotechnology Centre , DBSKKV, Dapoli
