Molecule lec 7
•Download as PPTX, PDF•
0 likes•90 views
Molecules can be classified based on the number of atoms they contain (monoatomic, diatomic, triatomic, polyatomic), the type of atoms (homoatomic containing the same type vs heteratomic containing different types), and their size (micro molecules are small while macromolecules are large, containing thousands of atoms). Common examples of different types of molecules include diatomic molecules like O2 and N2, heteratomic triatomic molecules like H2O and CO2, and macromolecules like proteins.
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Grade 8 Chemistry Structure of Matter : Atoms, Molecules and Ions

Grade 8 Chemistry Structure of Matter : Atoms, Molecules and Ions
Similar to Molecule lec 7
Similar to Molecule lec 7 (20)
General Biology. LECTURE 2. THE CHEMICAL FOUNDATION OF LIFE.pptx

General Biology. LECTURE 2. THE CHEMICAL FOUNDATION OF LIFE.pptx
namma_kalvi_11th_chemistry_unit_1_ppt_em_218218.pptx

namma_kalvi_11th_chemistry_unit_1_ppt_em_218218.pptx
Recently uploaded
Recently uploaded (20)
Recombinant DNA technology (Immunological screening)

Recombinant DNA technology (Immunological screening)
Pests of mustard_Identification_Management_Dr.UPR.pdf

Pests of mustard_Identification_Management_Dr.UPR.pdf
All-domain Anomaly Resolution Office U.S. Department of Defense (U) Case: “Eg...

All-domain Anomaly Resolution Office U.S. Department of Defense (U) Case: “Eg...
Kochi ❤CALL GIRL 84099*07087 ❤CALL GIRLS IN Kochi ESCORT SERVICE❤CALL GIRL

Kochi ❤CALL GIRL 84099*07087 ❤CALL GIRLS IN Kochi ESCORT SERVICE❤CALL GIRL
Pulmonary drug delivery system M.pharm -2nd sem P'ceutics

Pulmonary drug delivery system M.pharm -2nd sem P'ceutics
GUIDELINES ON SIMILAR BIOLOGICS Regulatory Requirements for Marketing Authori...

GUIDELINES ON SIMILAR BIOLOGICS Regulatory Requirements for Marketing Authori...
Hire 💕 9907093804 Hooghly Call Girls Service Call Girls Agency

Hire 💕 9907093804 Hooghly Call Girls Service Call Girls Agency
Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...

Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...
Formation of low mass protostars and their circumstellar disks

Formation of low mass protostars and their circumstellar disks
VIRUSES structure and classification ppt by Dr.Prince C P

VIRUSES structure and classification ppt by Dr.Prince C P
Nightside clouds and disequilibrium chemistry on the hot Jupiter WASP-43b

Nightside clouds and disequilibrium chemistry on the hot Jupiter WASP-43b
High Class Escorts in Hyderabad ₹7.5k Pick Up & Drop With Cash Payment 969456...

High Class Escorts in Hyderabad ₹7.5k Pick Up & Drop With Cash Payment 969456...
Molecule lec 7
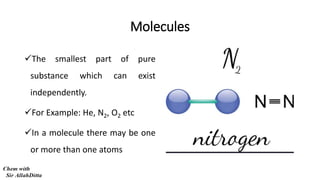
- 1. Molecules The smallest part of pure substance which can exist independently. For Example: He, N2, O2 etc In a molecule there may be one or more than one atoms
- 3. Classification of Molecules Molecule may be classified on the basis of: 1. Number of Atoms present in molecule 2. Type of Atoms present in molecule 3. Size of Atom present in molecule
- 4. 1. Number of Atoms. Mono atomic • The molecules which consist of only one atom are called Mono atomic Molecules • For example: He, Ne, Ar , etc. • Note: Only Noble gasses are Monoatomic molecules in UNIVERSE.
- 5. Di-atomic Molecules The molecules which are consist of two atoms are called di- atomic molecules For example: H2 ,O2 , N2, Cl2 etc. Molecules are formed for the sake of Compeletion duplets and octets of the atoms.
- 6. Tri-atomic Molecules • The molecules which are consist of three atoms are called tri- atomic molecules. • For example: CO2, O3, H2O etc.
- 7. Polyatomic Molecules • The molecules which are consist of many atoms are called Polyatomic molecules • For example: C6H12O6, H2SO4
- 8. Type of Atoms On the Basis of type of atoms molecules may be have two types 1. Homo-atomic 2. Hetro-atomic
- 9. Homo & Hetro Atomic Molecules The molecules which are consist of same type of atoms are called homoatomic molecules For Example H2, N2, Cl2 etc. The molecules which are consist of different type of atoms are called hetroatomic molecules For example: H2O, CO2, etc.
- 10. Micro and Macro Molecules On the basis of Size molecule may have two types 1. Micro molecules 2. Macromolecules
- 11. Micro Molecules The molecules which are small in size and have low molecular weight are called micro molecules. For example CO2, CH4, etc. All the molecules which we consider in chemistry are almost micromolecules almost
- 12. Macro Molecules The molecules which are larger in size and have high molecular weights are called macromolecules. For Example. haemoglobin, Steroids, Carbohydrates, etc. There are 10 thousand atoms in haemoglobin and it is 68 thousand times heavier than Hydrogen atom
- 13. Atomicity
- 16. Questions. What are molecules? Give examples Differentiate between micro and macro molecules What are homo and hetro molecules? Give examples. What are Mono di and tri atomic molecules? give examples of each.