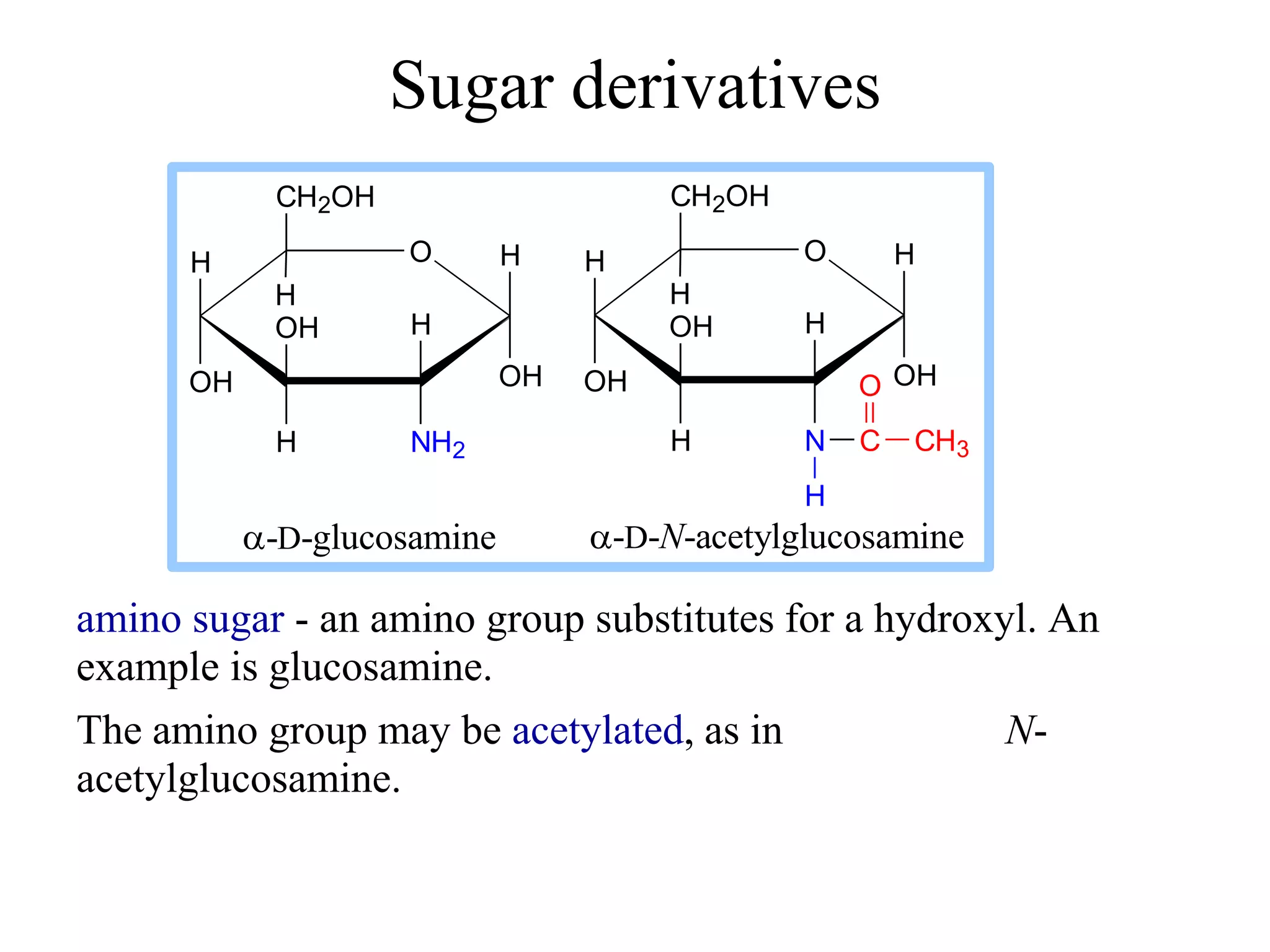
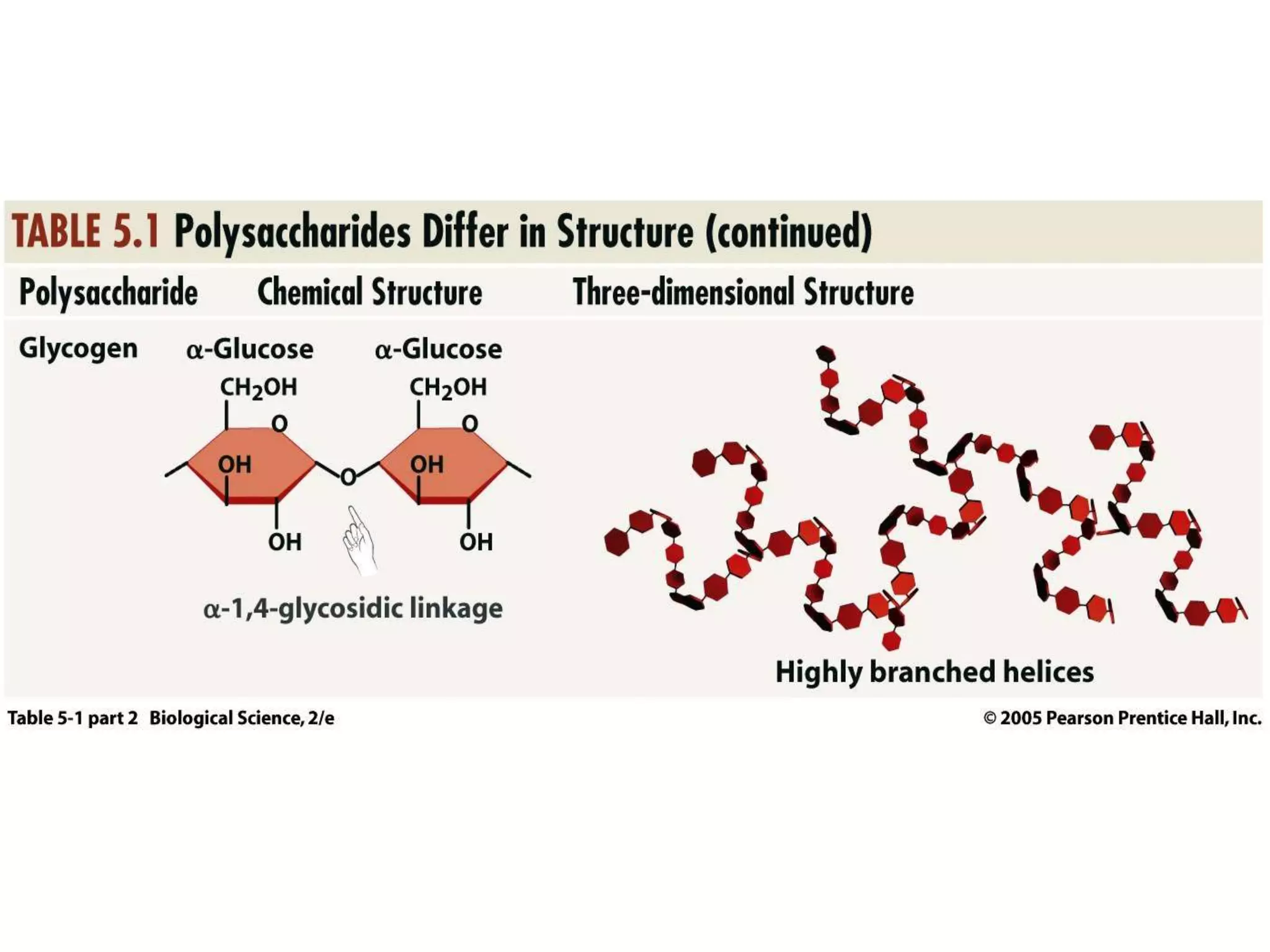
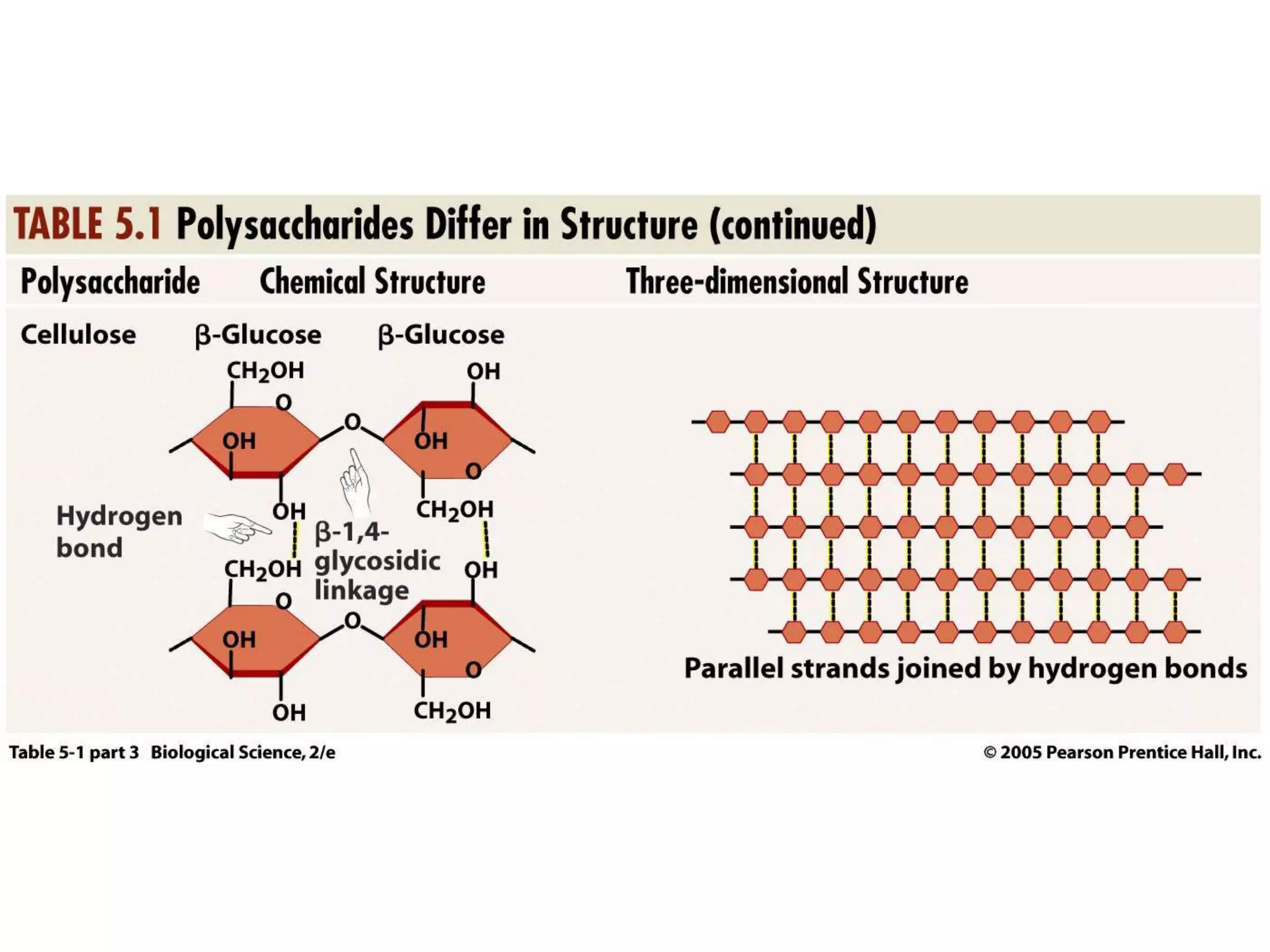
This document discusses the modification and derivatives of carbohydrates. It describes sugar alcohols, acids, and amino sugars as sugar derivatives. It then covers glycosidic bonds in disaccharides like maltose and cellobiose. Polysaccharides discussed include starch, glycogen, and cellulose. Glycosaminoglycans and proteoglycans are also summarized. Finally, it briefly mentions O-linked oligosaccharides on glycoproteins.