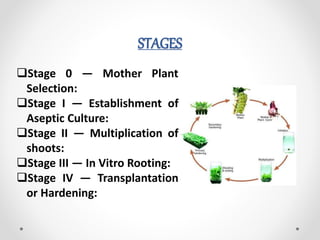
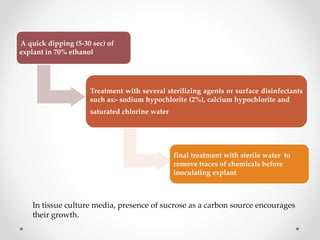
Micropropagation is a technology for rapidly producing many plant clones in limited space. The process involves several stages: selecting a disease-free mother plant, establishing aseptic cultures, multiplying shoots, rooting plantlets, and hardening before transplantation. This method allows for disease-free plant production, conservation of endangered species, and efficient plant material exchange.