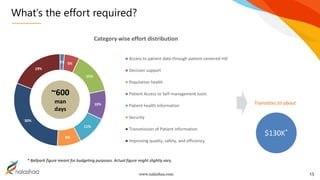
The document outlines the Meaningful Use Stage 3 (MU3) implementation by a software services company specializing in technology solutions. It discusses various requirements and changes associated with MU3, including data capture, application access, and reporting changes, emphasizing the need for a collaborative approach for certification. Finally, it addresses the effort required for compliance and the timeline implications for EHR vendors as they prepare for MU3 adoption.