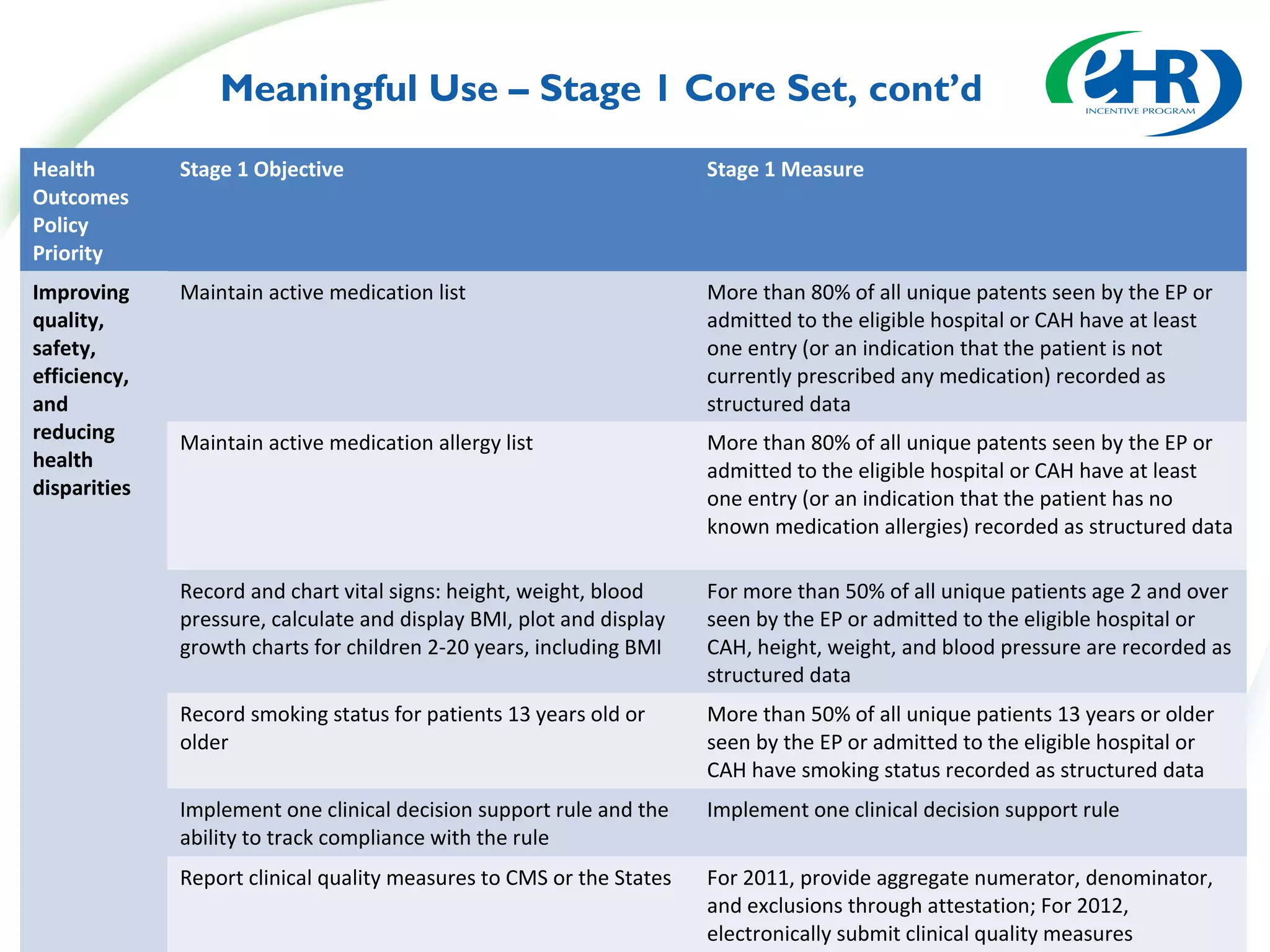
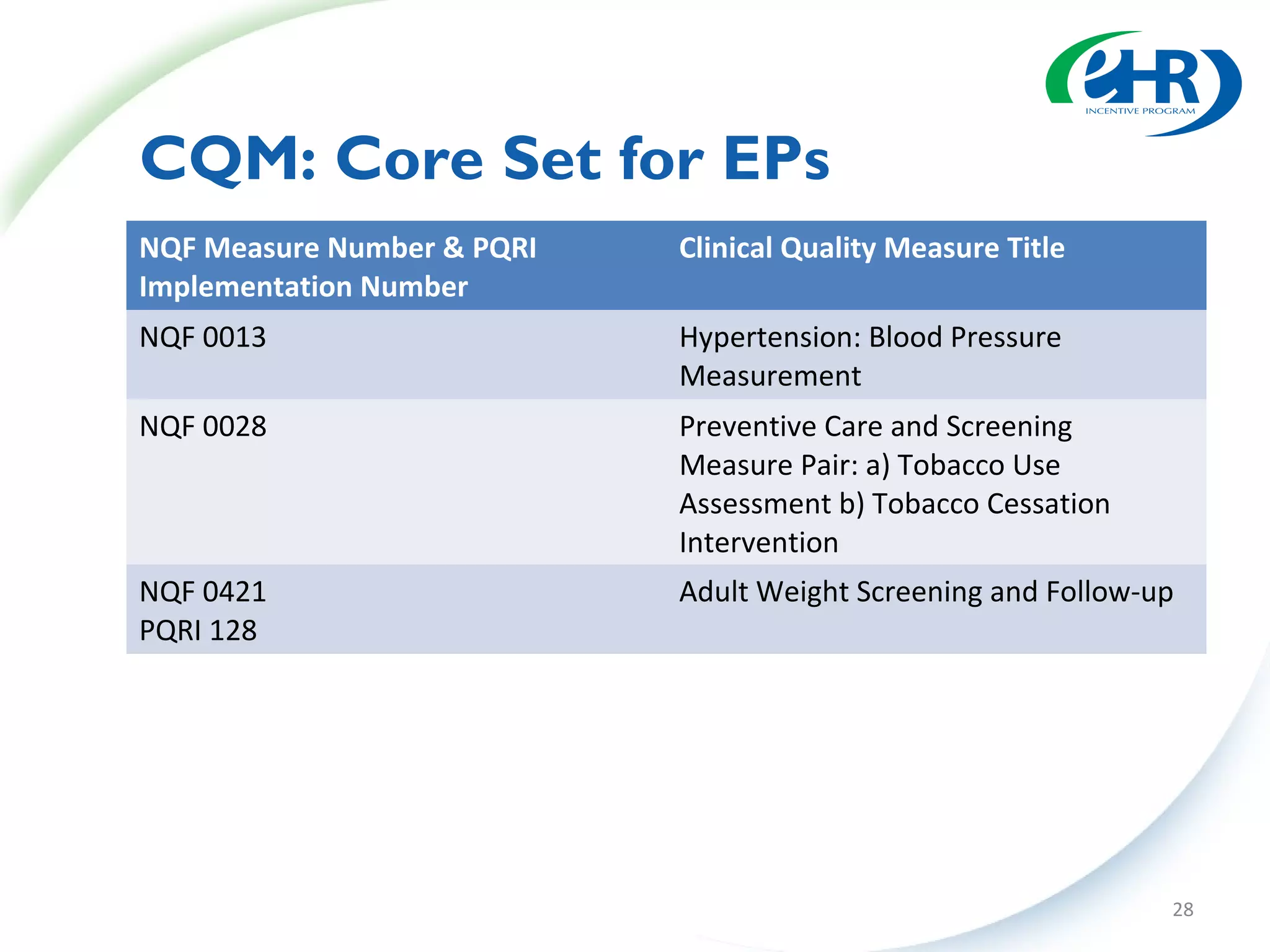
The document discusses changes between the proposed rule and final rule for implementing the Medicare and Medicaid EHR Incentive Program under the HITECH Act. Key changes included lowering the thresholds for meaningful use measures, modifying clinical quality measures, and clarifying eligible provider and hospital definitions. The final rule aimed to ease burden and address concerns raised during public comment period.