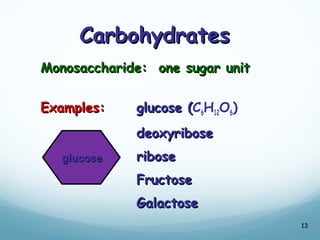
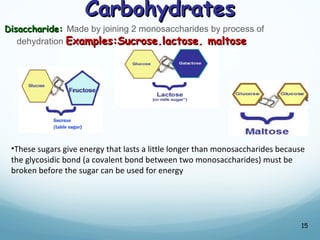
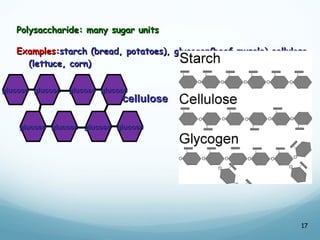
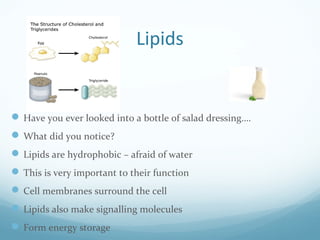
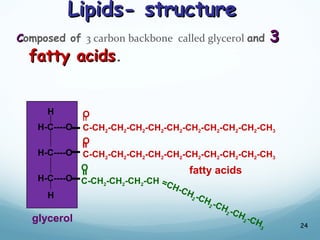
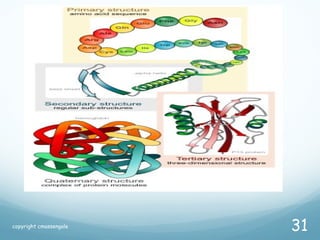
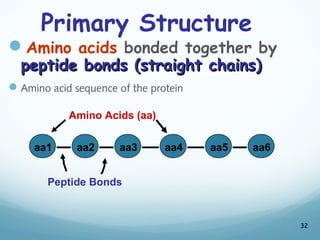
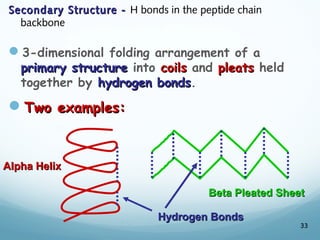
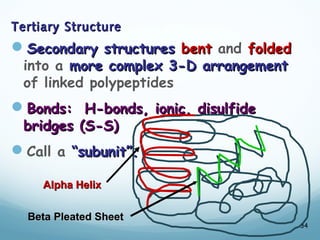
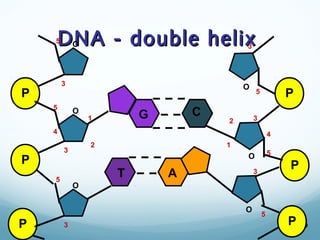
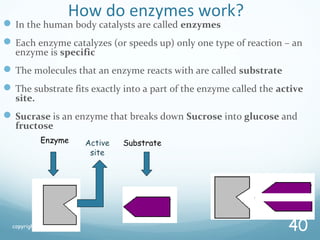
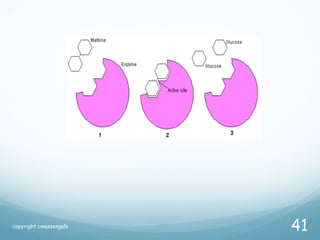
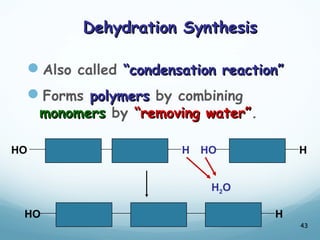
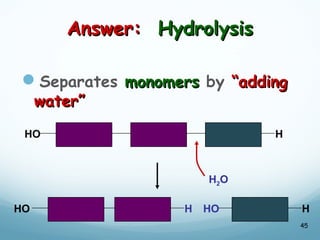
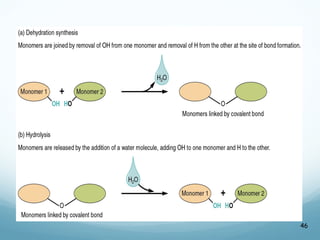
All living things are primarily composed of large biomolecules called biomolecules, which are made up of many atoms bonded together. Biomolecules contain carbon and are classified into four main types: carbohydrates, lipids, proteins, and nucleic acids. Carbohydrates include sugars and starches, lipids are fats and oils, proteins are made of amino acids, and nucleic acids include DNA and RNA. These macromolecules are essential for life and perform important functions in cells and organisms.