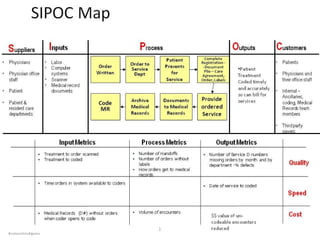
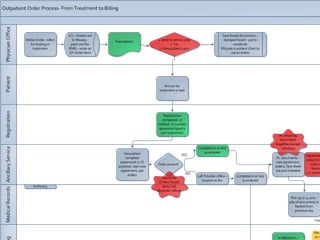
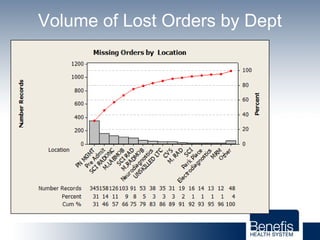
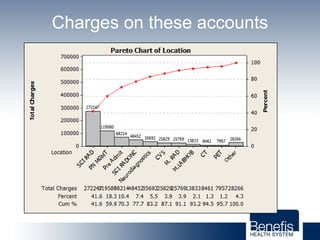
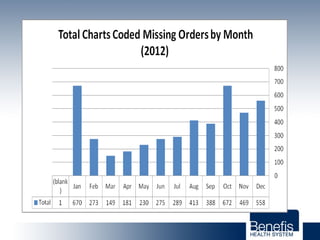
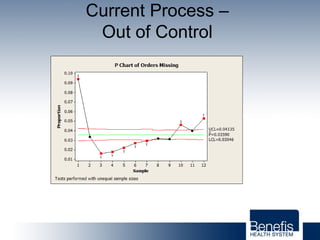
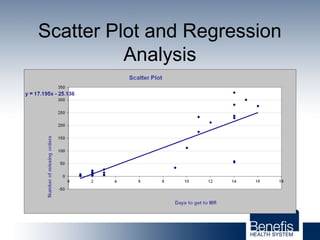
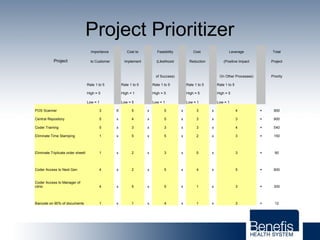
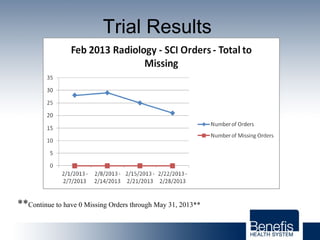
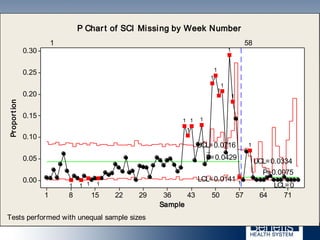
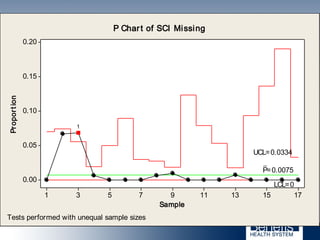
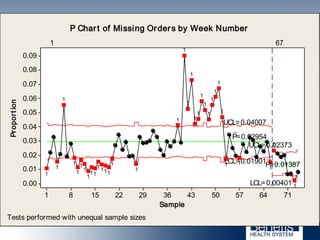
Based in Great Falls, Montana, Benefis Health System serves over 230,000 people across a 15-county region. With over 2,600 employees, Benefis is the largest non-governmental employer in the Great Falls area. The organization operates two campuses with a total of 516 beds and partners with more than 250 local physicians.