This document provides an overview of a chemical process control course. It includes:
- A list of course policies including exams, assignments, and grading.
- An outline of topics to be covered in the course ranging from process modeling to controller design.
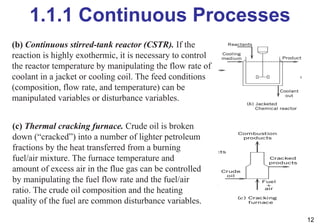
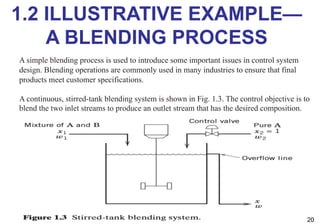
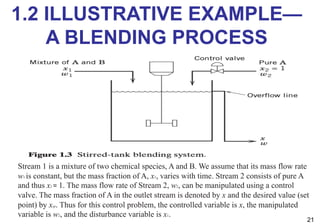
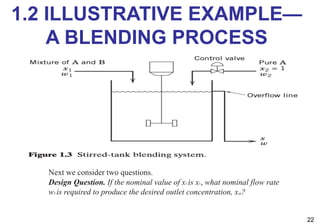
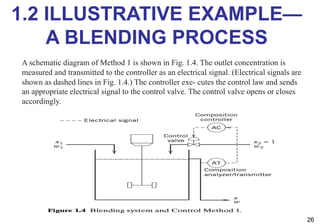
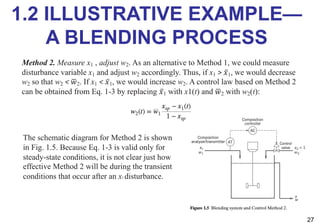
- An introduction to process control concepts including different types of processes (continuous, batch, semibatch), control variables, and strategies for controlling processes like a blending operation.
The document provides details on the structure, content, and objectives of the chemical process control course.