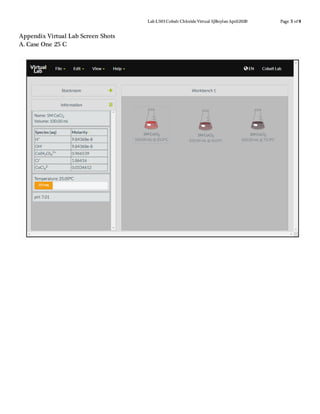
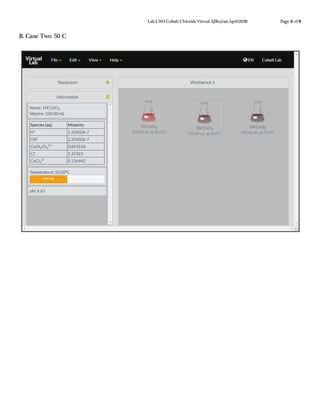
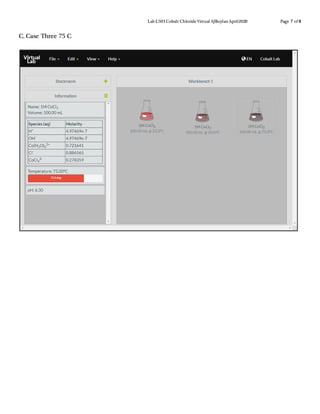
This document describes a virtual lab experiment to estimate the equilibrium constant, Gibbs free energy, enthalpy and entropy of cobalt chloride solutions at different temperatures. Students are instructed to use an online simulation to collect concentration and equilibrium constant data for cobalt chloride solutions at 25°C, 50°C, and 75°C. They then calculate additional thermodynamic values like Gibbs free energy, enthalpy and entropy using the collected data and appropriate equations like Cramer's rule and van't Hoff analysis. The goal is to determine these important thermodynamic properties of the cobalt chloride system at varying temperatures.



![Lab L503 Cobalt ChlorideVirtual SJBoylanApril2020 Page 4 of 8
18. Use Cramers rule to calculate the entropy
(G3x(-E1) - G1x(-E3) / (1x(-E1) - 1x(E3)) => I
19. Use the van’t Hoff equation two point form to calculate the enthalpy
ln(K3 / K1) = (delta H / R)x (1/T1 - 1/T3)
delta H = 8.31/1000 x ln(K3 / K1) / (1/T1 - 1/T3)
20. The reaction is
Co(H2O)6 2+ (aq) + 4 Cl- (aq) < == > CoCl4 2- (aq) + 6 H2O
21. The equilibrium expression is
Kcomplex ion = [CoCl4 2-] / ( [Co(H2O)6 2+} x [Cl-]^4)](https://image.slidesharecdn.com/l503labvirtualcobaltchloride-200408175532/85/L503-lab-virtual-cobalt-chloride-4-320.jpg)