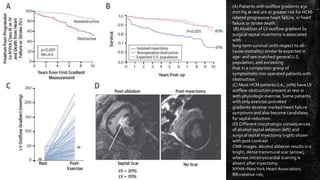
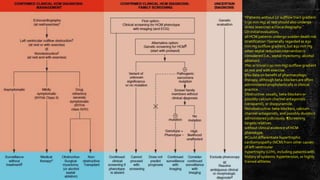
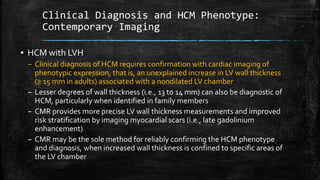
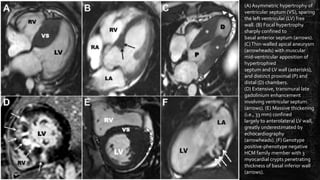
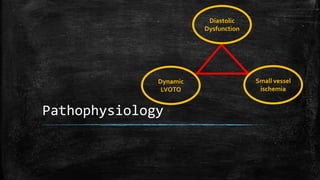
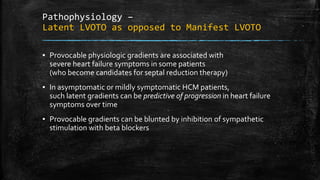
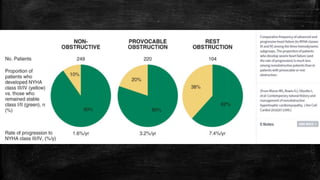
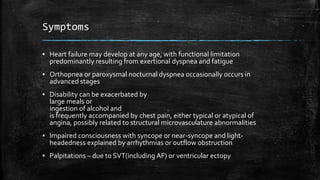
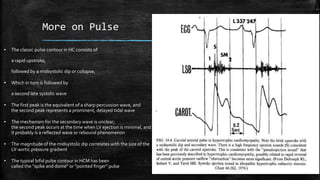
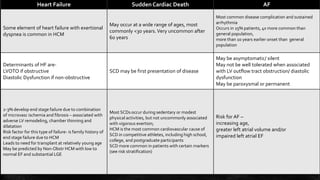
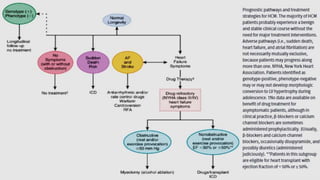
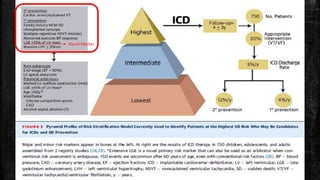
Hypertrophic cardiomyopathy (HCM) is a common inherited cardiac disease characterized by left ventricular thickening, with various genetic mutations identified as primary causes. The document discusses genetic screening, clinical features, diagnosis, and the natural history of HCM, including risk stratification for sudden cardiac death. It emphasizes the importance of imaging techniques and genetic testing in managing the disease and screening family members for potential risks.






















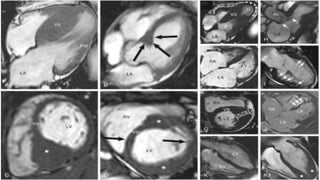












































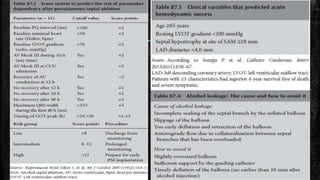
![Heart Failure With Obstruction -
Invasive Treatment
▪ Because of the duration of experience, documented long-term results, and
safety data
Surgical septal myectomy is considered the preferred treatment for patients
with advanced limiting HF symptoms due to outflow gradients of ≥ 50 mm
Hg (at rest and/or with physiologic [exercise] provocation)
who are refractory to maximal medical management
▪ Long-term studies over 40 years in HCM have shown that
surgical myectomy reliably reverses HF symptoms by permanently
abolishing obstruction,
restoring normal LV pressures, and reducing or abolishing mitral
regurgitation
▪ Operative mortality for septal myectomy is now <1%](https://image.slidesharecdn.com/hypertrophiccardiomyopathy-200526142109/85/Hypertrophic-cardiomyopathy-84-320.jpg)