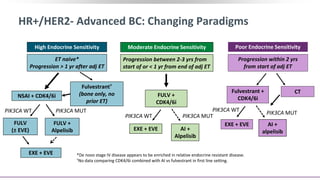
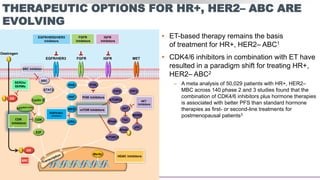
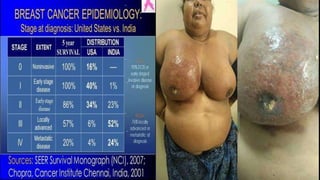
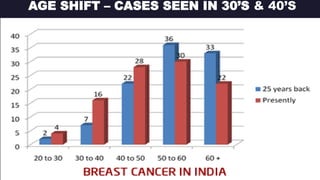
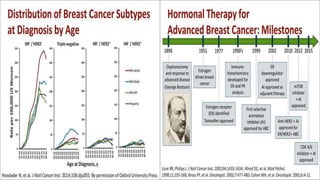
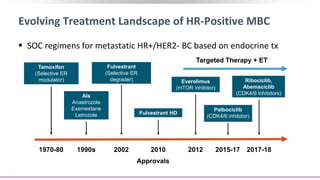
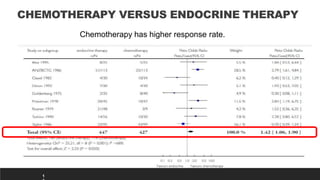
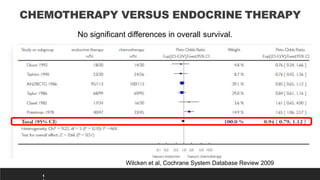
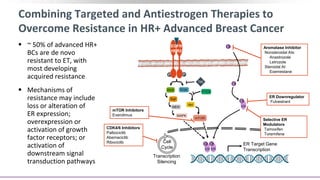
This presentation discusses breast cancer, specifically hormone receptor positive/HER2 negative breast cancer. It provides statistics on the distribution of breast cancer molecular subtypes in the United States. It also discusses the evolving treatment landscape for hormone receptor positive metastatic breast cancer, including the approvals and use of targeted therapies in combination with endocrine therapy, such as CDK4/6 inhibitors. Clinical trial results are summarized that demonstrate improved progression-free survival when a CDK4/6 inhibitor is added to first-line endocrine therapy for advanced hormone receptor positive breast cancer. An approach to personalized therapy is proposed based on factors such as endocrine sensitivity and molecular alterations.



![G2
S
M
G1
pRB
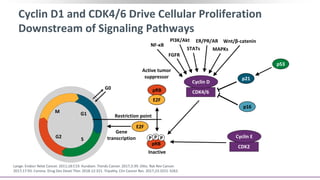
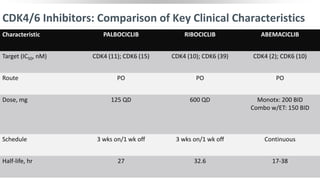
The Role of CDK4/6 in Breast Cancer
Growth of HR+ MBC is
dependent on cyclin D1, a direct
transcriptional target of ER
Cyclin D1 activates CDK4/6,
resulting in G1-S phase
transition and cell cycle entry[1]
Some cell-line models of
endocrine resistance show
dependence on cyclin D1 and
CDK4/6[2,3]
ERα Mitogenic
signaling
S phase transcription
program
G1/S transition
P P P
pRB
E2F
E2F
CDK1
Cyclin B
CDK1/2
Cyclin A
CDK2
Cyclin E
CDK4/6
Cyclin D](https://image.slidesharecdn.com/hrher2neumbc-210321143728/85/Hr-her2-neu-mbc-13-320.jpg)

![20
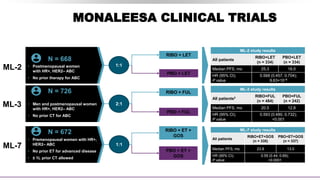
MONARCH CLINICAL TRIALS
Footer text here. Arial 7pt font in black.
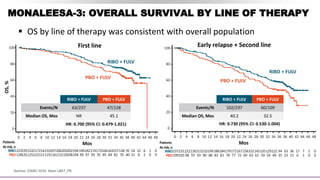
MONARCH-2
MONARCH-3
• Pre- and postmenopausal
women with HR+,
HER2– ABC
• Progression on ET
([neo]adjuvant or 1L
advanced)
N = 669
1:1
ABE + FUL
PBO + FUL
MONARCH-2 study results
All patients
ABE+FUL
n = 446
PBO+FUL
n = 223
Median PFS, mo 16.4 9.3
HR (95% CI);
P value
0.553 (0.449, 0.681);
<0.001
N = 493
1:1
ABE + NSAI
PBO + NSAI
MONARCH-3 study results
All patients
ABE+NSAI
n = 328
PBO+NSAI
n = 165
Median PFS, mo NR 14.7
HR (95% CI);
P value
0.54 (0.41, 0.72);
0.000021
N = 493
• Postmenopausal women
with HR+, HER2– ABC
• No prior systemic therapy
in the advanced setting](https://image.slidesharecdn.com/hrher2neumbc-210321143728/85/Hr-her2-neu-mbc-20-320.jpg)

![Approach to Therapy for HR+/HER2- MBC:
Move to Personalization
1L[1-4] 2L[1] 3L
AI + CDK4/6i Fulvestrant ±
everolimus
Exemestane +
everolimus[8]
Fulvestrant + CDK4/6i
Exemestane +
everolimus
4L/5L[1]
Taxane or
capecitabine
4L+[1]
Eribulin
PIK3CAm:
fulvestrant +
alpelisib[5]
BRCAm: olaparib or
talazoparib[9,10]
Sacituzumab
govitecan
(IMMU-132)[13]
ESR1m: SERD (+ CDK4/6i)[6,7]
HER2 low: trastuzumab
deruxtecan
(DS8201a)[11]
HER2m: neratinib[12]](https://image.slidesharecdn.com/hrher2neumbc-210321143728/85/Hr-her2-neu-mbc-23-320.jpg)