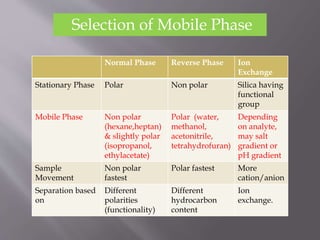
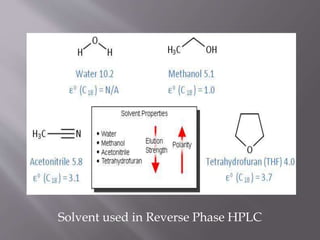
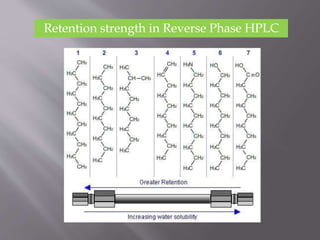
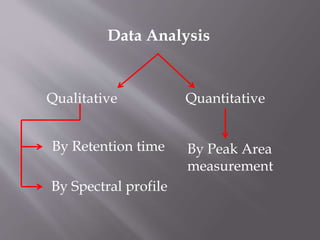
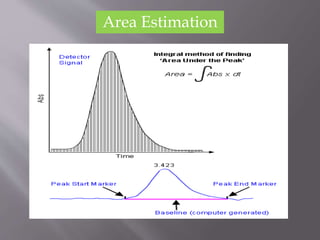
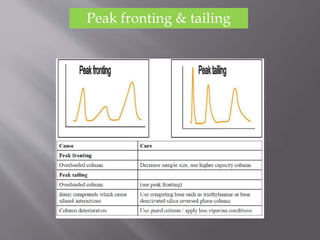
The document outlines the process of HPLC method development, focusing on the selection of stationary and mobile phases based on molecular characteristics of the sample, including charge, hydrophobicity, and solubility. It also details data analysis techniques for qualitative and quantitative assessment, utilizing retention time and spectral profiles for peak identification. Troubleshooting common issues such as unusual peak shapes and fluctuating retention times is also discussed.