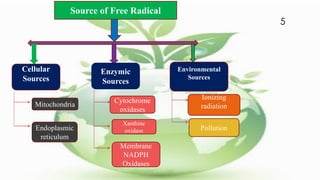
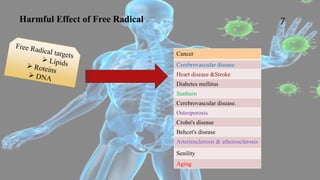
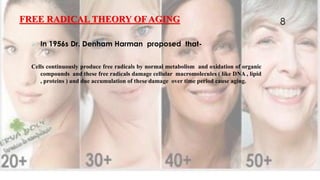
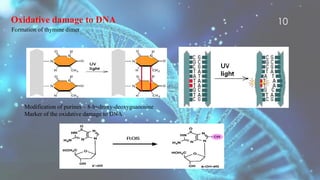
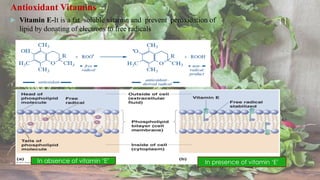
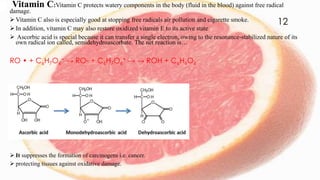
Free radicals are unstable molecules that can cause harm through reactions with other compounds. They are produced through normal cellular processes but also environmental sources like radiation and pollution. Free radicals can damage DNA, proteins, and lipids, potentially contributing to aging and diseases. Antioxidants like vitamins C and E can help defend against free radical damage by donating electrons to the free radicals to make them stable. The free radical theory of aging proposes that accumulation of free radical damage to cellular components over time is a major factor in the aging process.