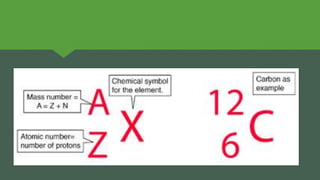
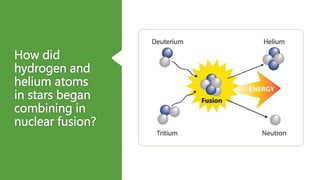
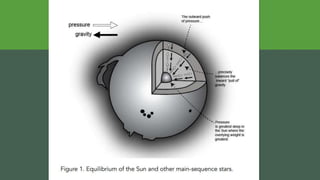
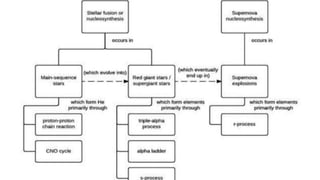
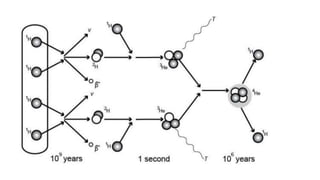
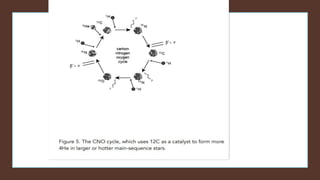
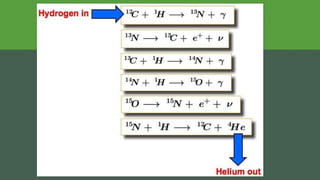
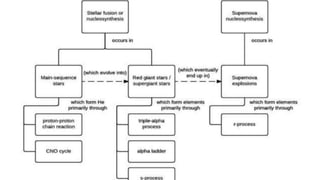
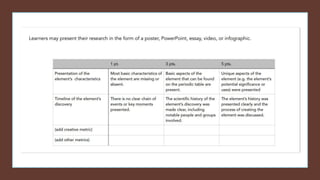
The document discusses how heavier elements are formed in stars through nuclear fusion reactions. It begins by outlining the key terms and concepts, including fusion, stellar nucleosynthesis, and the proton-proton chain reaction. It then explains several important nuclear fusion processes that occur in stars, such as the triple alpha process, alpha ladder, CNO cycle, and how these lead to the formation of carbon, oxygen, and heavier elements up to iron. The document also describes stages in the life of stars, from main sequence stars to red giants and supernovae, which generate even heavier elements through rapid neutron capture processes.