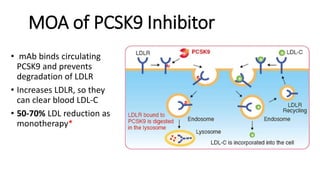
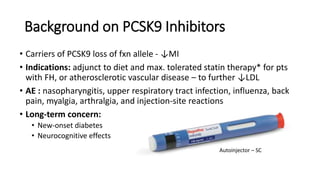
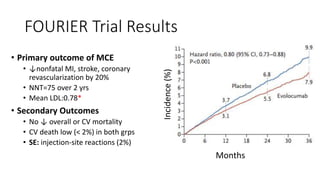
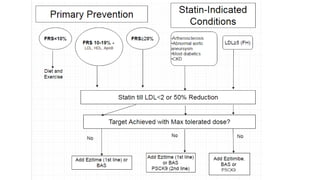
This document summarizes the cardiovascular benefits of evolocumab, a PCSK9 inhibitor, based on the FOURIER trial. The FOURIER trial found that adding evolocumab to moderate or high-intensity statin therapy reduced major cardiovascular events like heart attack, stroke, and coronary revascularization by 20% over 2 years compared to placebo in high-risk patients. Evolocumab lowered LDL cholesterol by 60% on average and had an acceptable safety profile with only injection site reactions occurring in more than 2% of patients. While evolocumab did not reduce overall or cardiovascular mortality, it provides an effective additional option to further lower LDL and reduce cardiovascular risk when added to statin therapy.