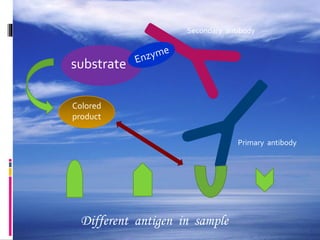
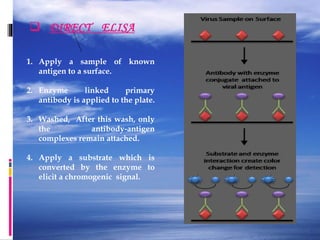
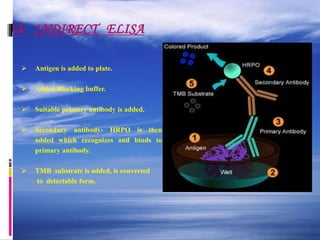
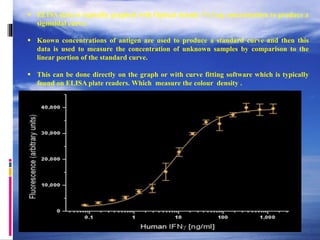
The document provides a comprehensive overview of the enzyme-linked immunosorbent assay (ELISA), detailing its principle, history, and various types including direct, indirect, and sandwich ELISA. ELISA is a sensitive technique primarily used for detecting and quantifying antigens or antibodies in samples, often applied in medical diagnostics, such as determining hormone levels and screening for viral infections. The document emphasizes the methodology, advantages, and disadvantages of each ELISA type while including essential reagents and applications.