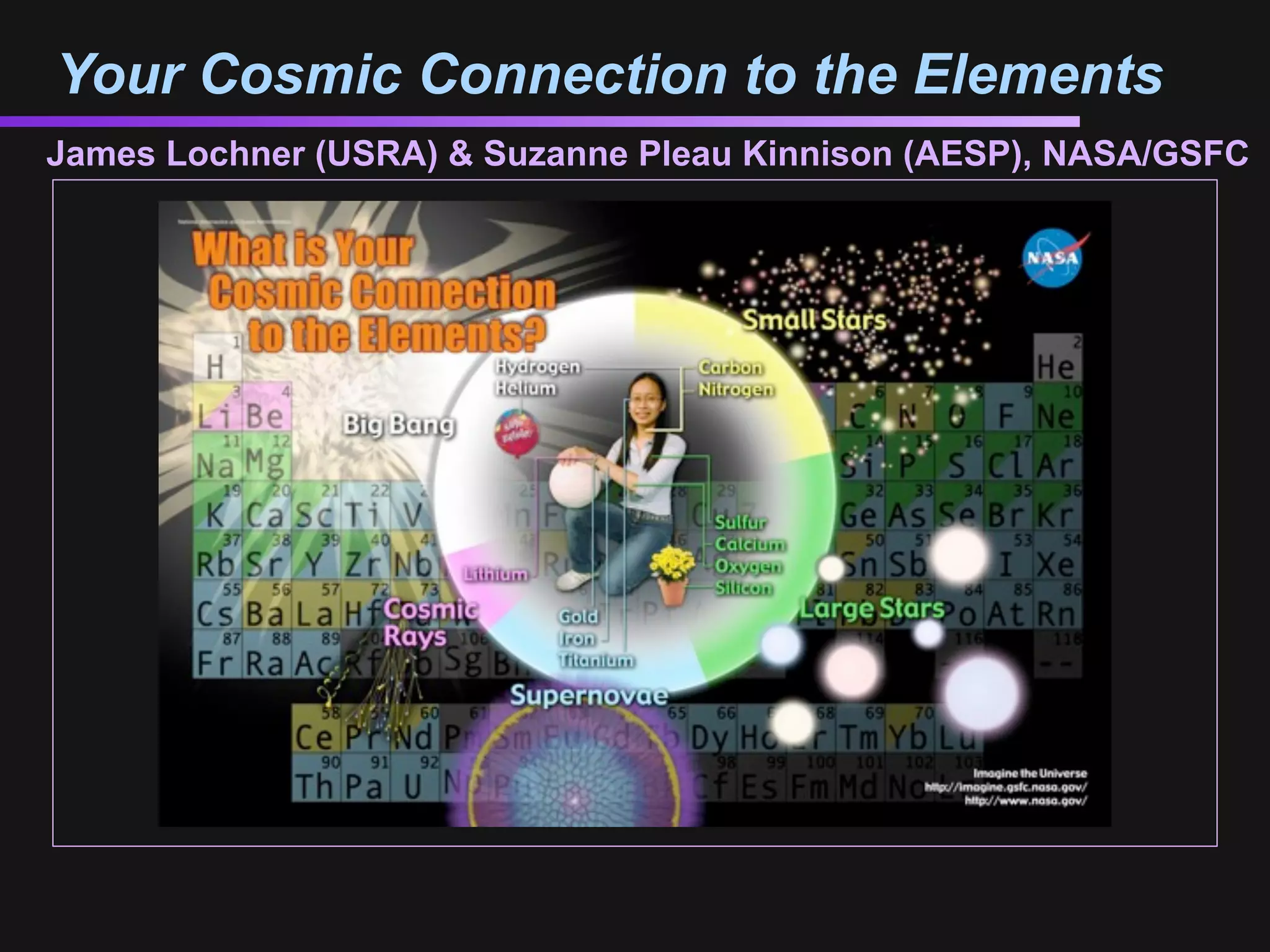
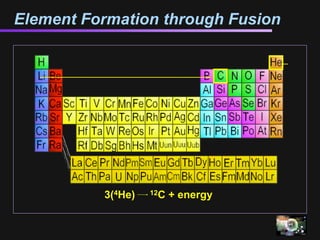
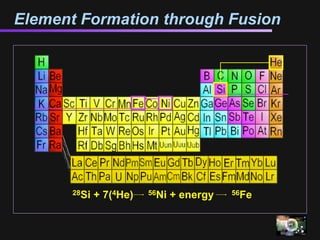
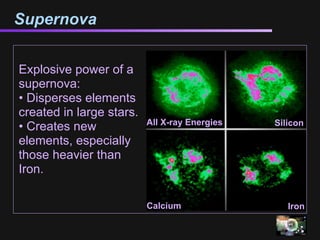
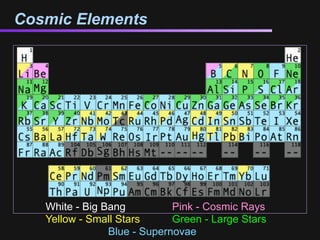
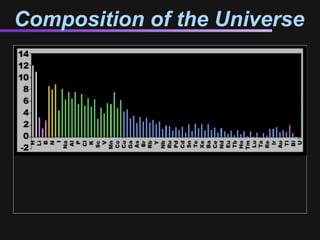
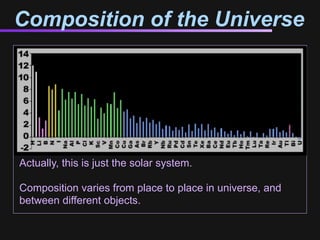
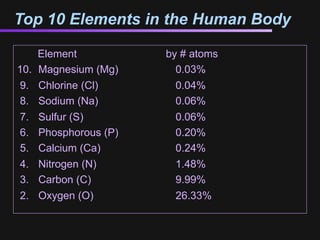
The document discusses how all the elements were formed in the universe. It explains that shortly after the Big Bang, the first light elements like hydrogen and helium formed. Later, in the cores of stars, nuclear fusion processes fused these light elements into heavier elements up to iron. The most massive stars ended their lives as supernovae, which fused even heavier elements and dispersed them throughout the universe. Some rare light elements like lithium and beryllium were produced through cosmic ray bombardment of interstellar gas. All elements found on Earth and in our bodies were originally produced in earlier generations of stars and supernovae.