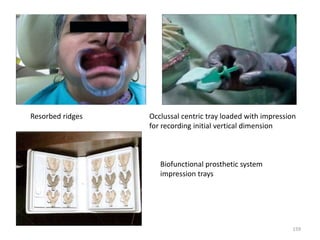
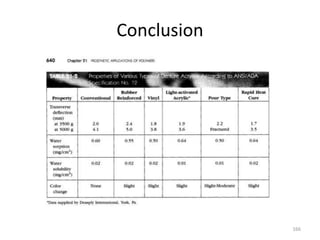
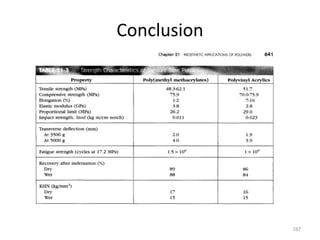
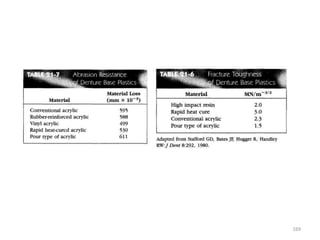
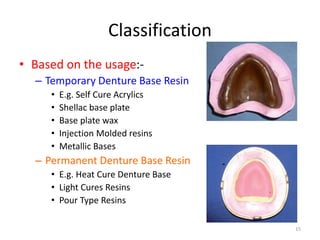
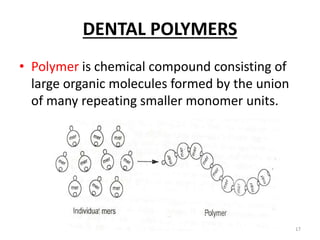
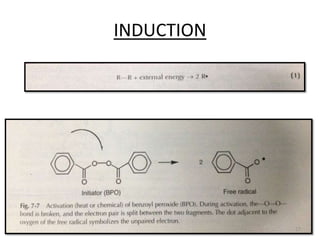
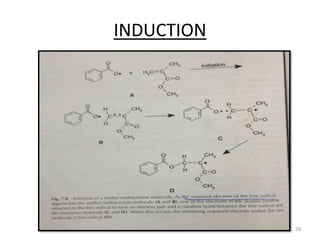
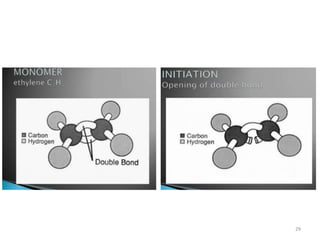
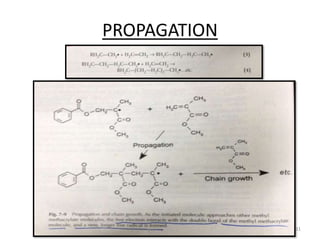
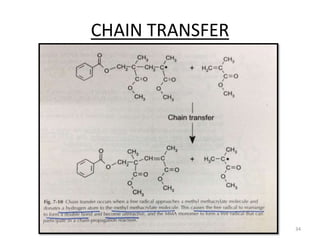
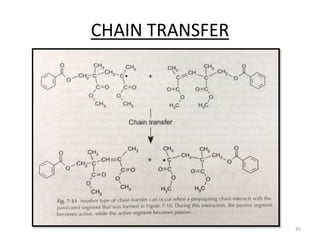
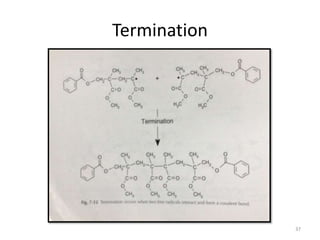
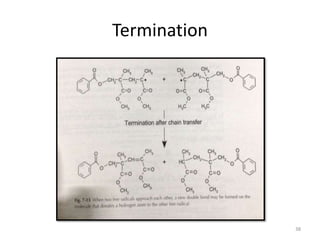
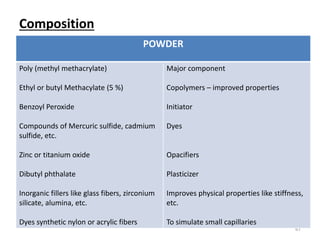
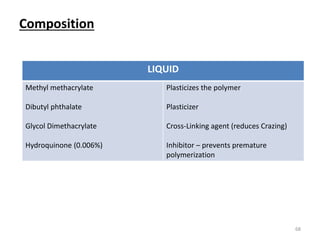
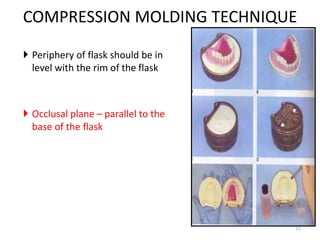
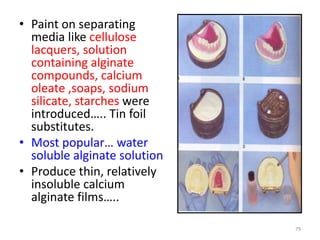
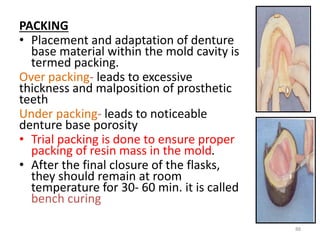
The document provides an overview of denture base resins including their definition, history, classification, key ingredients, and properties. It discusses the early use of materials like ivory, bone, and porcelain for dentures and the later development of vulcanite in the 1840s as the first affordable and workable material. Polymethyl methacrylate (PMMA) was introduced in the 1930s and became the standard material by 1946, providing improved properties over previous materials. The document outlines the polymerization process and ideal requirements for denture base resins based on biocompatibility, durability, and other factors. Heat-cured PMMA denture base resin is currently the most widely used material.
































































![• First reported by Kimura et. al
• This technique employs a
specially formulated resin and
a non metallic flasks. FRP Flask
[ Fiber Reinforced Plastic flasks]
95
POLYMERIZATION VIA MICROWAVE
ENERGY](https://image.slidesharecdn.com/msbpl7jssjmew2s8nwpj-signature-11bfc635dadd10a3a09f0305d36d2634b5c4d35ec2e6d3761fabe8171afe7f12-poli-160505064925/85/Denture-Base-Resin-95-320.jpg)

![• No noticeable difference
• Trade name
– Keystone Diamond D
• Disadvantages
Flasks are expensive and have tendency to
break down after processing several dentures.
The polycarbon bolts tend to break if
tightened too firmly.
97
Bernard Levin et al [ JPD 1989;61: 381-383]](https://image.slidesharecdn.com/msbpl7jssjmew2s8nwpj-signature-11bfc635dadd10a3a09f0305d36d2634b5c4d35ec2e6d3761fabe8171afe7f12-poli-160505064925/85/Denture-Base-Resin-97-320.jpg)





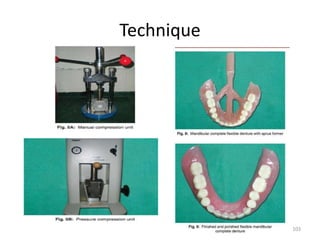











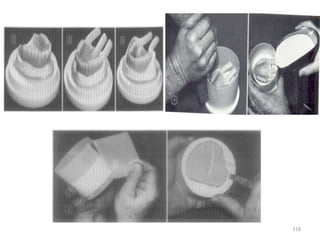
![Fluid Resin Technique
• Employs a pourable chemically activated
resin
Advantages
• Improved adaptation to underlying soft
tissues.
• Decreased damage to prosthetic teeth and
denture base during deflasking.
• Reduced material costs.
• Simplification of flasking, deflasking, finishing
procedure
115
Walter Shepard [ JPD 1968;19: 562-564]](https://image.slidesharecdn.com/msbpl7jssjmew2s8nwpj-signature-11bfc635dadd10a3a09f0305d36d2634b5c4d35ec2e6d3761fabe8171afe7f12-poli-160505064925/85/Denture-Base-Resin-115-320.jpg)
![Fluid Resin Technique
Disadvantages
• Noticeable shifting of prosthetic teeth
during processing
• Air entrapment
• Poor bonding between denture base and
acrylic teeth
• Technique sensitivity
116
Walter Shepard [ JPD 1968;19: 562-564]](https://image.slidesharecdn.com/msbpl7jssjmew2s8nwpj-signature-11bfc635dadd10a3a09f0305d36d2634b5c4d35ec2e6d3761fabe8171afe7f12-poli-160505064925/85/Denture-Base-Resin-116-320.jpg)
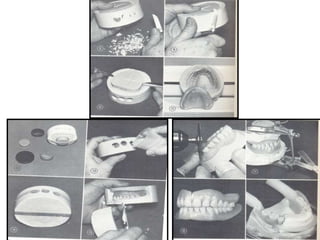
![• Fluid denture resin processing in a rigid mold
• Koblitz FF et al described a fluid resin processing
technique using rigid, modified gypsum
investment as replacement for hydrocolloid
investment.
• Advantages
• Method requires no specialized equipment such
as metal flasks or hydrocolloid conditioning
apparatus
• The technique eliminates the time consuming
step of sorting and replacing artificial teeth in as
hydrocolloid mold.
117[ JPD 1973; 30; 339-345 ]](https://image.slidesharecdn.com/msbpl7jssjmew2s8nwpj-signature-11bfc635dadd10a3a09f0305d36d2634b5c4d35ec2e6d3761fabe8171afe7f12-poli-160505064925/85/Denture-Base-Resin-117-320.jpg)







![Modulus of elasticity
• Resins have sufficient stiffness [ 2400MPa] for
use in complete and partial dentures.
Impact Strength
• It is the measure of energy absorbed by a
material when it is broken by a sudden blow.
• Addition of plasticizers increase the impact
strength.
129](https://image.slidesharecdn.com/msbpl7jssjmew2s8nwpj-signature-11bfc635dadd10a3a09f0305d36d2634b5c4d35ec2e6d3761fabe8171afe7f12-poli-160505064925/85/Denture-Base-Resin-129-320.jpg)














![Cytotoxicity of Denture Base Acrylic
Resins
[ JPD 2003: 90; 190-195 ]
• Residual monomer, resulting from incomplete conversion of
monomers into polymer, has the potential to cause
– irritation,
– inflammation,
– and an allergic responses of oral mucosa.
• Clinical signs and symptoms reported include
– erythema,
– erosion of oral mucosa,
– burning sensation of mucosa and tongue.
148](https://image.slidesharecdn.com/msbpl7jssjmew2s8nwpj-signature-11bfc635dadd10a3a09f0305d36d2634b5c4d35ec2e6d3761fabe8171afe7f12-poli-160505064925/85/Denture-Base-Resin-148-320.jpg)
![Cytotoxicity of Denture Base Acrylic
Resins
[ JPD 2003: 90; 190-195 ]
Effect of polymer : monomer ratio
• More monomer added to the mixture, the greater amount of
residual monomer and therefore more potential for cytotoxicity
.
Effect of storage time and water immersion
• Sheridan et al reported that cytotoxic effect of acrylic resins was
greater in first 24 hours after polymerization and decreased with
time.
• Therefore it is recommended that dentist soak the resin
prosthesis in water for atleast 24 hours before placing them in
the patients mouth.
149](https://image.slidesharecdn.com/msbpl7jssjmew2s8nwpj-signature-11bfc635dadd10a3a09f0305d36d2634b5c4d35ec2e6d3761fabe8171afe7f12-poli-160505064925/85/Denture-Base-Resin-149-320.jpg)