Culture of Renal Proximal Tubule Epithelial Cell Line SA7K Using Extracellular Matrices Improved Functionality of Key Drug Transporters
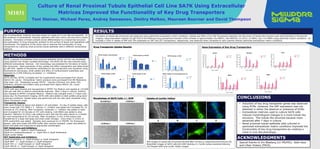
- 1. SA7K, a pseudo immortalized renal proximal epithelial tubule cell line was developed using Sigma’s Zinc Finger nuclease technology. The SA7K cell line was cultured using different extracellular matrices. Cell morphology was assessed for formation of tubule like structures. Gene expression of key uptake and efflux transporters was determined by qRT-PCR. Uptake of fluorescent substrates for key transporters was visualized using fluorescence microscopy, while uptake and efflux of nonfluorescent substrates was assessed by LC/MS following incubation +/- inhibitors. Materials: SA7K cell line, RPTEC Complete and Tox supplements were purchased from Sigma Aldrich (St. Louis). Extracellular matrix products were purchased from BD Bioscience (San Jose, CA). Substrates (except YM155 - Cayman Chemical, Ann Arbor, MI), inhibitors, solvents and buffers were purchased from Sigma Aldrich (St. Louis). Culture Conditions: SA7K cells were thawed and resuspended in RPTEC Tox Medium and seeded at 125,000 cells per well with or without extracellular matrices. After 2 days in culture, medium was changed to RPTEC Complete Medium. Medium was changed every 2-3 days until assay day. For fluorescent imaging, SA7K cells were plated on ibidi µslides using same culturing conditions. Uptake assay was performed and live cells were visualized using Nikon fluorescent scope. Transporter Assays: Cells were thawed as above and plated in 24 well plates. On day of uptake assay, cells were rinsed with 37C HBSS x 3. Vehicle +/- inhibitor was added and incubated for 10 minutes at 37C shaking. After incubation, substrate +/- inhibitor was added to wells and incubated for 10 minutes at 37C with shaking. Cold HBSS was added to stop reaction. Methanol (0.25mL) was added to each well and the plate placed on a shaker at room temperature for 20 minutes. After incubation, 0.2mL of the extract was transferred to a deep well plate and dried under nitrogen. Once dried, 0.125mL of 50% acetonitrile was added. Samples were analyzed by LC-MS/MS. For fluorescent assays, cells were lysed with RIPA buffer after reaction stopped. Lysate was added to black well plate and measured at appropriate wavelengths. OAT Substrates and Inhibitors: 10µM of PAH +/- 10µM or 25µM Probenecid 50µM of 6 carboxyfluorescein +/- 10µM PAH or 10µM Probenecid 50µM Lucifer Yellow OCT Substrates and Inhibitors: 10µM Amantadine +/- 10µM Doxepin or 10µM Verapamil 10µM MPP+ +/- 10µM Doxepin or 10µM Verapamil 10µM TEA +/- 10µM Doxepin or 10µM Verapamil 10µM YM155 +/- 10µM Doxepin or 10µM Verapamil METHODS Culture of Renal Proximal Tubule Epithelial Cell Line SA7K Using Extracellular Matrices Improved the Functionality of Key Drug Transporters Toni Steiner, Michael Perez, Andrey Samsonov, Dmitry Malkov, Maureen Bourner and David Thompson M1031 • Induction of key drug transporter genes was observed using ECMs. However, the OAT expression was not detected in either the absence or presence of ECMs. • Extracellular matrices used to culture SA7K cells induced morphological changes to a more tubule like structure. This tubule like structure became more advanced after 7 days in culture. • Renal proximal tubule epithelial cells cultured in optimized extracellular matrix conditions improved the functionality of key drug transporters by creating a more in-vivo like phenotype. CONCLUSIONS Use of extracellular matrices has been shown to create an in-vivo like environment. In the presence of extracellular matrices, epithelial cells form cell to cell and cell to matrix contacts. Formation of these contacts drives differentiation to a tubule like structure and polarization of drug transporter proteins to the surface of apical and basolateral cell membranes. The objective of this study was to optimize the functionality of drug transporters by culturing renal proximal tubule epithelial cells in different extracellular matrices. PURPOSE Gene Expression of Key Drug Transporters Formation of tubule like structures was observed using optimized extracellular matrix conditions. Uptake and efflux of an OAT fluorescent substrate into the lumen of tubule like structure was demonstrated by fluorescent imaging. Gene expression analysis by qRT-PCR of the following drug transporters showed a fold increase of approximately 20x MATE1, 50x MATE2K, 5x OCT2, 2x MRP2, and 1.5x MRP4 using optimized culture conditions. An increase ≥ 50% in uptake of PAH and TEA substrates by OAT and OCT transporters, respectively, was observed when compared to cells cultured without optimized extracellular matrix conditions. RESULTS OCT Substrates OAT Substrates Uptake of Lucifer Yellow A B C D 40X images of tubule like structures formed after 10 days in optimized ECM cultures (A) Brightfield images of SA7K cells (B) DAPI staining (C) Lucifer yellow substrate staining (D) Merged DAPI and Lucifer Yellow images. Drug Transporter Uptake Results Morphology of SA7K Cells +/- ECM No ECM Day 7 + ECM Day 1 + ECM Day 4 + ECM Day 7 Special thanks to Jim Blasberg (LC-MS/MS), Kelly Keys and Lillian Vickery (FACS). ACKNOWLEDGMENTS