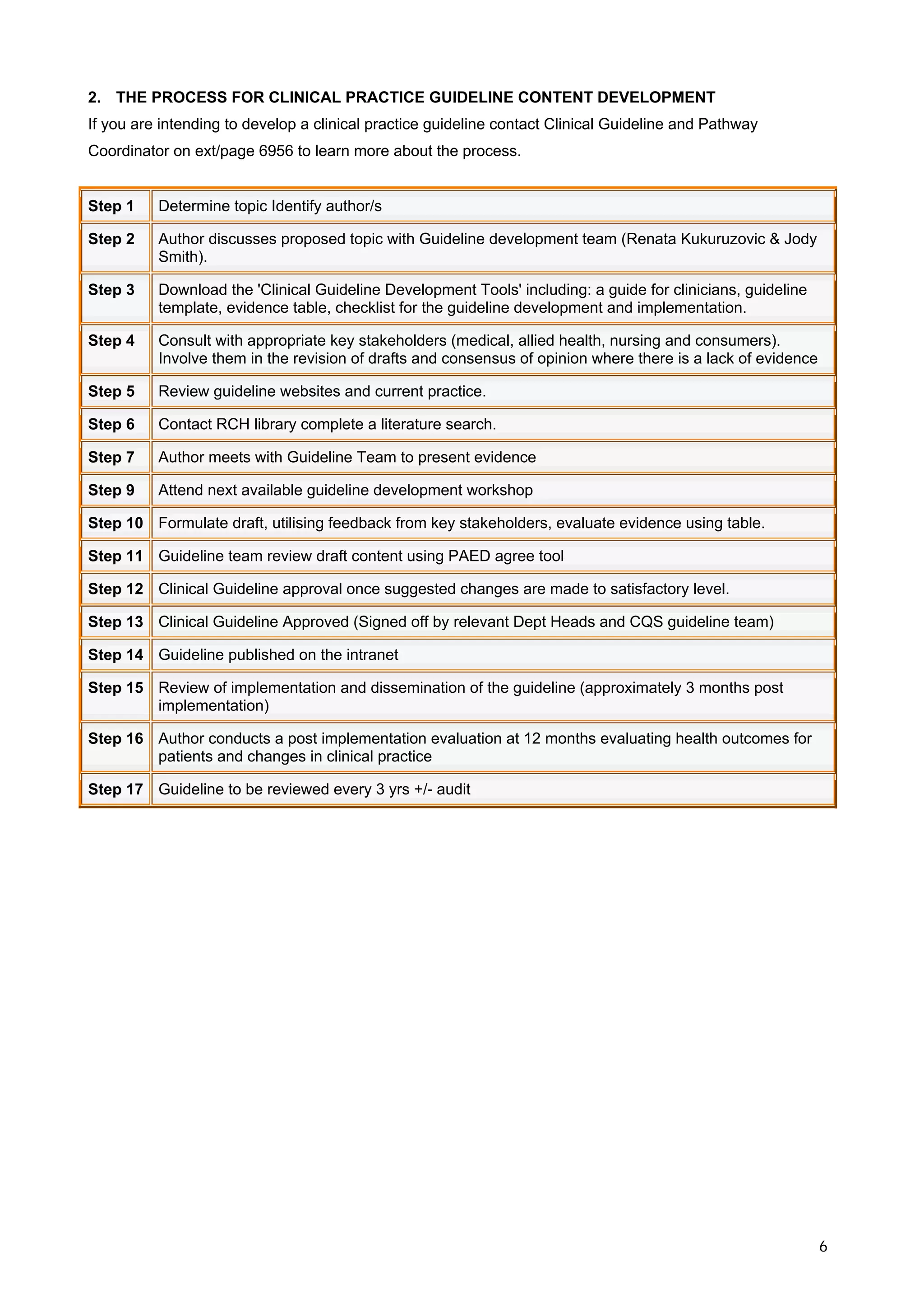
This document provides guidance for developing clinical practice guidelines at the Royal Children's Hospital in Melbourne, Australia. It outlines a 17 step process for guideline development that involves identifying a topic, forming an authoring team, reviewing evidence, drafting content, obtaining stakeholder feedback, finalizing and approving the guideline, implementing it, and evaluating its impact. Key principles include developing guidelines through a multidisciplinary process, basing them on the best available research evidence, and involving consumers throughout. The overall goal is to improve healthcare quality and outcomes for patients.