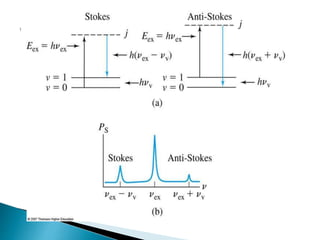
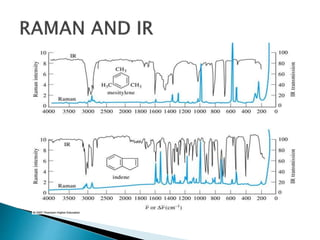
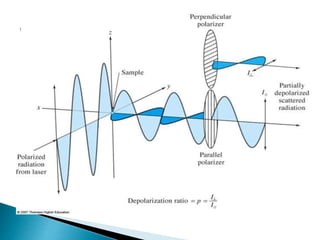
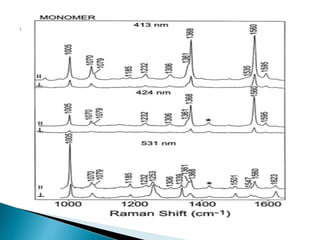
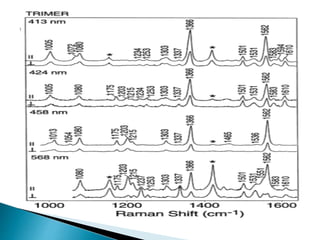
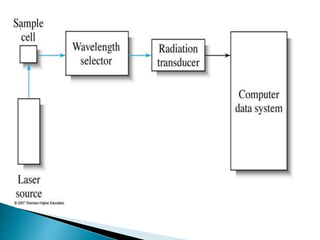
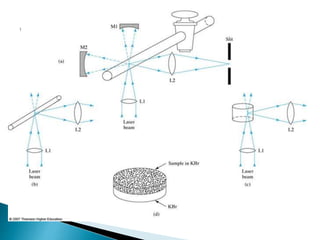
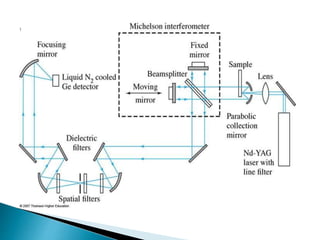
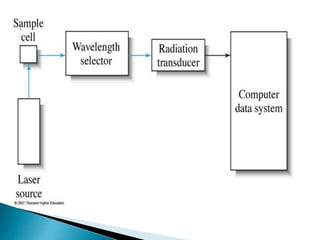
Raman spectroscopy and infrared spectroscopy are similar techniques for analyzing molecular vibrations, but they differ in their operating principles. Raman spectroscopy analyzes the scattering of monochromatic light, such as a laser, while infrared spectroscopy analyzes light absorption. Raman spectroscopy can be used to analyze aqueous solutions because water is a weak Raman scatterer, whereas water strongly absorbs infrared light. Both techniques provide information about molecular structure through vibrational fingerprints, but Raman spectroscopy has advantages for certain applications due to its ability to analyze solutions and its high sensitivity.