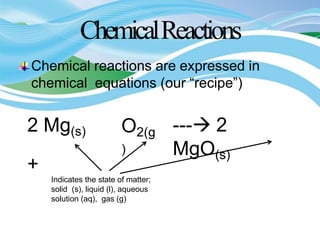
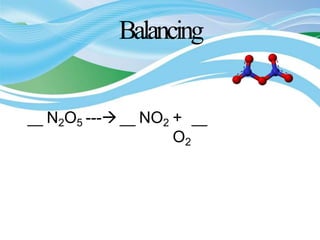
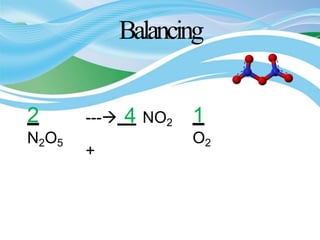
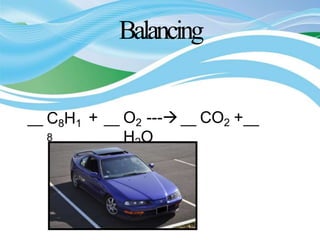
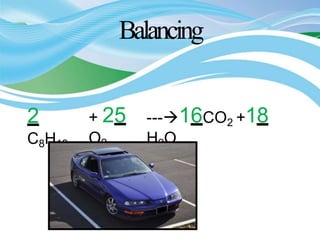
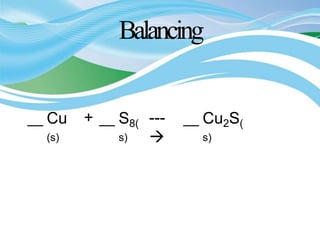
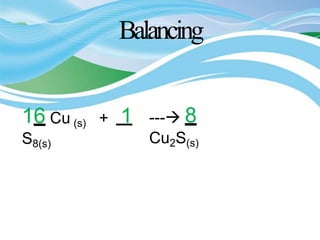
Stoichiometry is the study of quantitative relationships in chemical formulas and reactions. It allows chemists to calculate amounts of reactants and products using moles, molar mass, and balanced chemical equations. Chemical reactions have fixed ratios of reactants and products that can be used to determine limiting reactants and calculate mass changes. Sample problems demonstrate how stoichiometric calculations are used to find amounts of substances involved in chemical reactions.


![Stoichiometry
Stoicheion [Gr. “element” or “part”]
Metron [Gr. “measure”]
Study of quantitative aspects of
chemical formulas and reactions](https://image.slidesharecdn.com/chemistrystoichiometry-200828050319-1-230301234409-871abb9b/85/chemistrystoichiometry-200828050319-1-pptx-3-320.jpg)










![TheMole(mol)
SI unit for amount of substance
Defined as the amount of a substance that
contains the same number of entities as there
are atoms in exactly 12 grams of carbon-12.
That is
6.022 x1023 [called Avogrado’s number]
∴1 mol of carbon-12 contains 6.022
x1023 carbon-12 atoms
1 mol of H2O contains 6.022 x1023 H2O
molecules](https://image.slidesharecdn.com/chemistrystoichiometry-200828050319-1-230301234409-871abb9b/85/chemistrystoichiometry-200828050319-1-pptx-15-320.jpg)














![Sources
• Dragon Cave Holiday Cooking 2012. [Photos]
• Silberberg, Martin S. “Stoichiometry of Formulas and
Equations”. Principles of General Chemistry.
McGraw-Hill, 2010.](https://image.slidesharecdn.com/chemistrystoichiometry-200828050319-1-230301234409-871abb9b/85/chemistrystoichiometry-200828050319-1-pptx-37-320.jpg)