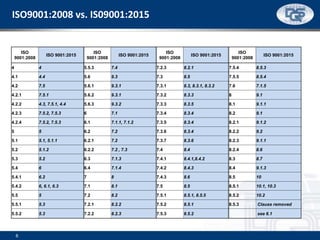
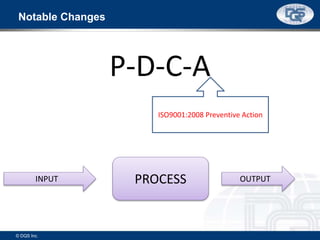
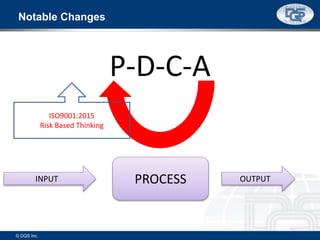
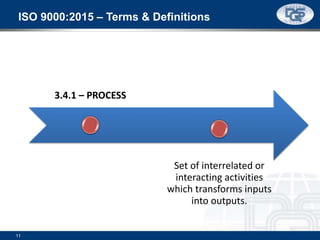
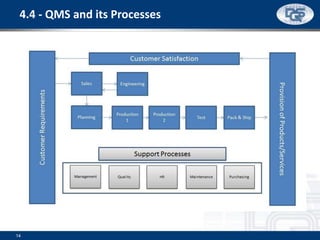
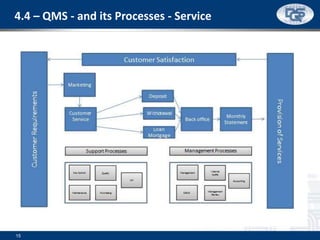
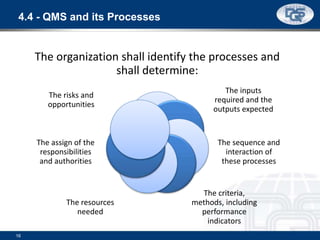
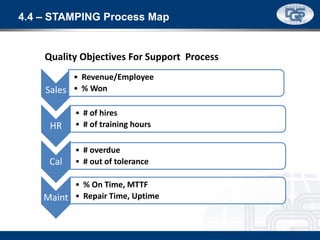
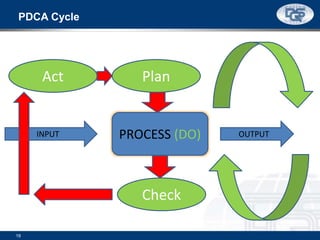
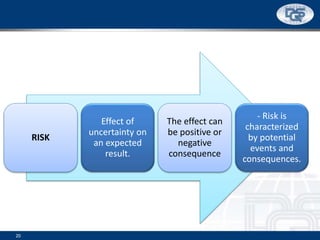
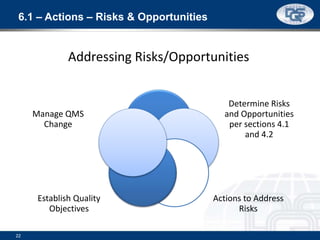
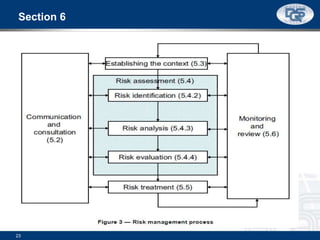
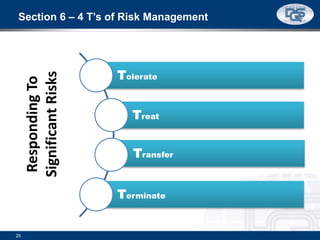
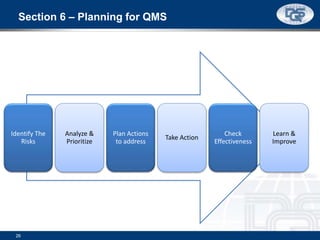
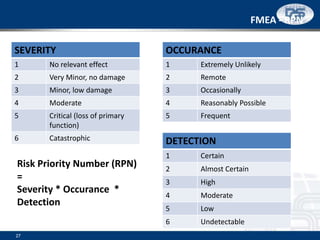
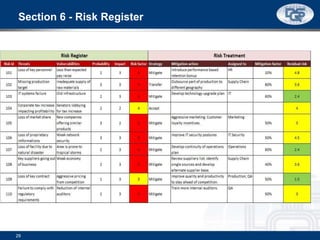
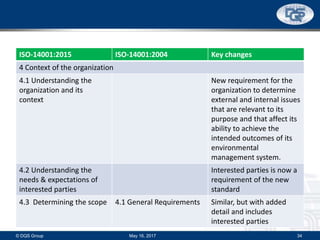
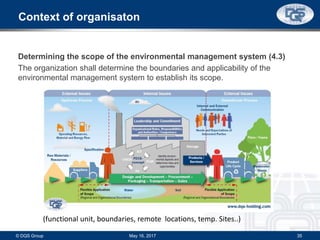
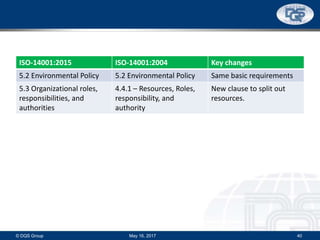
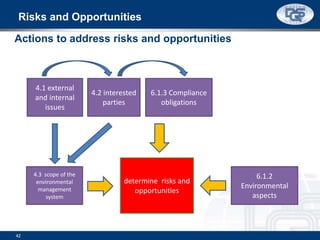
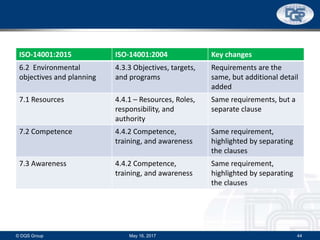
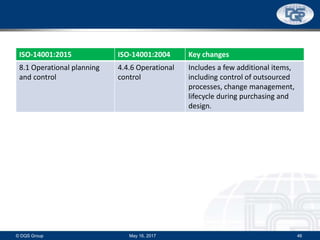
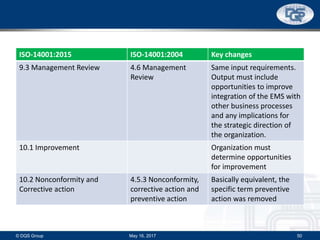
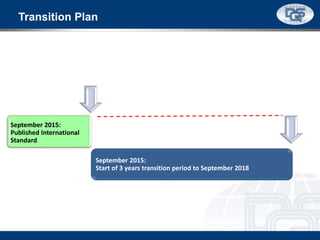
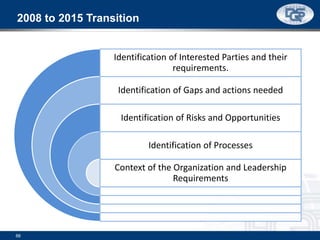
This document provides an overview of ISO 9001:2015 and ISO 14001:2015 standards, detailing key changes and requirements in areas such as quality management and environmental management systems. It emphasizes the importance of leadership commitment, risk management, and understanding organizational context in both standards. Additionally, it outlines the process approach, highlighting the benefits of process alignment and effectiveness to achieve desired outcomes.