Ch 323 INFARED SPECTROSCOPY LECTURE 2
•
0 likes•23 views
UNIVERSITY OF DAR ES SALAAM
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
Similar to Ch 323 INFARED SPECTROSCOPY LECTURE 2
Similar to Ch 323 INFARED SPECTROSCOPY LECTURE 2 (13)
Go with the most obvious one first...Carboxylic Acids will have a.pdf

Go with the most obvious one first...Carboxylic Acids will have a.pdf
12 infraredspectroscopyandmassspectrometry-wade7th-140409023806-phpapp01

12 infraredspectroscopyandmassspectrometry-wade7th-140409023806-phpapp01
12 - Infrared Spectroscopy and Mass Spectrometry - Wade 7th

12 - Infrared Spectroscopy and Mass Spectrometry - Wade 7th
Recently uploaded
Ultrasound color Doppler imaging has been routinely used for the diagnosis of cardiovascular diseases, enabling real-time flow visualization through the Doppler effect. Yet, its inability to provide true flow velocity vectors due to its one-dimensional detection limits its efficacy. To overcome this limitation, various VFI schemes, including multi-angle beams, speckle tracking, and transverse oscillation, have been explored, with some already available commercially. However, many of these methods still rely on autocorrelation, which poses inherent issues such as underestimation, aliasing, and the need for large ensemble sizes. Conversely, speckle-tracking-based VFI enables lateral velocity estimation but suffers from significantly lower accuracy compared to axial velocity measurements.
To address these challenges, we have presented a speckle-tracking-based VFI approach utilizing multi-angle ultrafast plane wave imaging. Our approach involves estimating axial velocity components projected onto individual steered plane waves, which are then combined to derive the velocity vector. Additionally, we've introduced a VFI visualization technique with high spatial and temporal resolutions capable of tracking flow particle trajectories.
Simulation and flow phantom experiments demonstrate that the proposed VFI method outperforms both speckle-tracking-based VFI and autocorrelation VFI counterparts by at least a factor of three. Furthermore, in vivo measurements on carotid arteries using the Prodigy ultrasound scanner demonstrate the effectiveness of our approach compared to existing methods, providing a more robust imaging tool for hemodynamic studies.
Learning objectives:
- Understand fundamental limitations of color Doppler imaging.
- Understand principles behind advanced vector flow imaging techniques.
- Familiarize with the ultrasound speckle tracking technique and its implications in flow imaging.
- Explore experiments conducted using multi-angle plane wave ultrafast imaging, specifically utilizing the pulse-sequence mode on a 128-channel ultrasound research platform. (May 9, 2024) Enhanced Ultrafast Vector Flow Imaging (VFI) Using Multi-Angle ...

(May 9, 2024) Enhanced Ultrafast Vector Flow Imaging (VFI) Using Multi-Angle ...Scintica Instrumentation
Recently uploaded (20)
Genome Projects : Human, Rice,Wheat,E coli and Arabidopsis.

Genome Projects : Human, Rice,Wheat,E coli and Arabidopsis.
The Mariana Trench remarkable geological features on Earth.pptx

The Mariana Trench remarkable geological features on Earth.pptx
LUNULARIA -features, morphology, anatomy ,reproduction etc.

LUNULARIA -features, morphology, anatomy ,reproduction etc.
Site specific recombination and transposition.........pdf

Site specific recombination and transposition.........pdf
Kanchipuram Escorts 🥰 8617370543 Call Girls Offer VIP Hot Girls

Kanchipuram Escorts 🥰 8617370543 Call Girls Offer VIP Hot Girls
GBSN - Microbiology (Unit 3)Defense Mechanism of the body 

GBSN - Microbiology (Unit 3)Defense Mechanism of the body
FAIRSpectra - Enabling the FAIRification of Analytical Science

FAIRSpectra - Enabling the FAIRification of Analytical Science
Selaginella: features, morphology ,anatomy and reproduction.

Selaginella: features, morphology ,anatomy and reproduction.
Role of AI in seed science Predictive modelling and Beyond.pptx

Role of AI in seed science Predictive modelling and Beyond.pptx
Porella : features, morphology, anatomy, reproduction etc.

Porella : features, morphology, anatomy, reproduction etc.
Cot curve, melting temperature, unique and repetitive DNA

Cot curve, melting temperature, unique and repetitive DNA
POGONATUM : morphology, anatomy, reproduction etc.

POGONATUM : morphology, anatomy, reproduction etc.
Efficient spin-up of Earth System Models usingsequence acceleration

Efficient spin-up of Earth System Models usingsequence acceleration
(May 9, 2024) Enhanced Ultrafast Vector Flow Imaging (VFI) Using Multi-Angle ...

(May 9, 2024) Enhanced Ultrafast Vector Flow Imaging (VFI) Using Multi-Angle ...
Ch 323 INFARED SPECTROSCOPY LECTURE 2
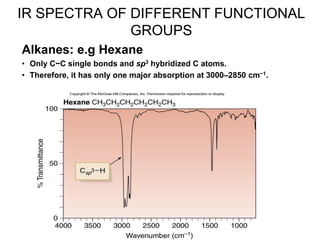
- 1. IR SPECTRA OF DIFFERENT FUNCTIONAL GROUPS Alkanes: e.g Hexane • Only C−C single bonds and sp3 hybridized C atoms. • Therefore, it has only one major absorption at 3000–2850 cm−1.
- 2. ALKENES e.g 1-Hexene • 1-Hexene has a C=C and Csp2−H, in addition to sp3 hybridized C atoms. • Therefore, there are three major absorptions: Csp2−H at 3150−3000 cm−1; Csp3−H at 3000−2850 cm−1; C=C at 1650 cm−1.
- 3. ALKYNES Absorptions from nitriles are generally of medium intensity and are clearly defined. Alkynes absorb weakly in this region unless they are highly asymmetric; NOTE: symmetrical alkynes do not show absorption bands. e.g.1-Hexyne • has a CC and Csp−H, in addition to sp3 hybridized C atoms. • Therefore, there are three major absorptions: Csp−H at 3300 cm−1; Csp3−H at 3000−2850 cm−1; CC at 2250 cm−1.
- 4. C N C≡C and C≡N Absorption The CC and CN triple bond absorptions at 2100- 2260 cm-1 are small but exposed/unique.
- 6. 6 Aromatic C-H and C=C O • Aromatic C-H around 3050 cm-1 and C=C two medium bands at about 1500 and 1600 cm-1.
- 7. ALCOHOLS
- 8. 8 Carboxylic Group O-H • Characteristic broad band (3400 – 2500 cm-1) due to H-bonding. • Identifies/confirms presence of a carboxylic acid.
- 9. ALDEHYDES
- 10. KETONES
- 11. AMINES
- 12. 12
- 13. 13 Base Values for IR Absorptions Bond type Wavenumbers O-H 3400 N-H 3400 C-H 3000 C≡N 2250 C≡C 2150 C=O 1715 C=C 1650 C-O 1100
- 15. IR-spec Problem
- 16. More Problems
