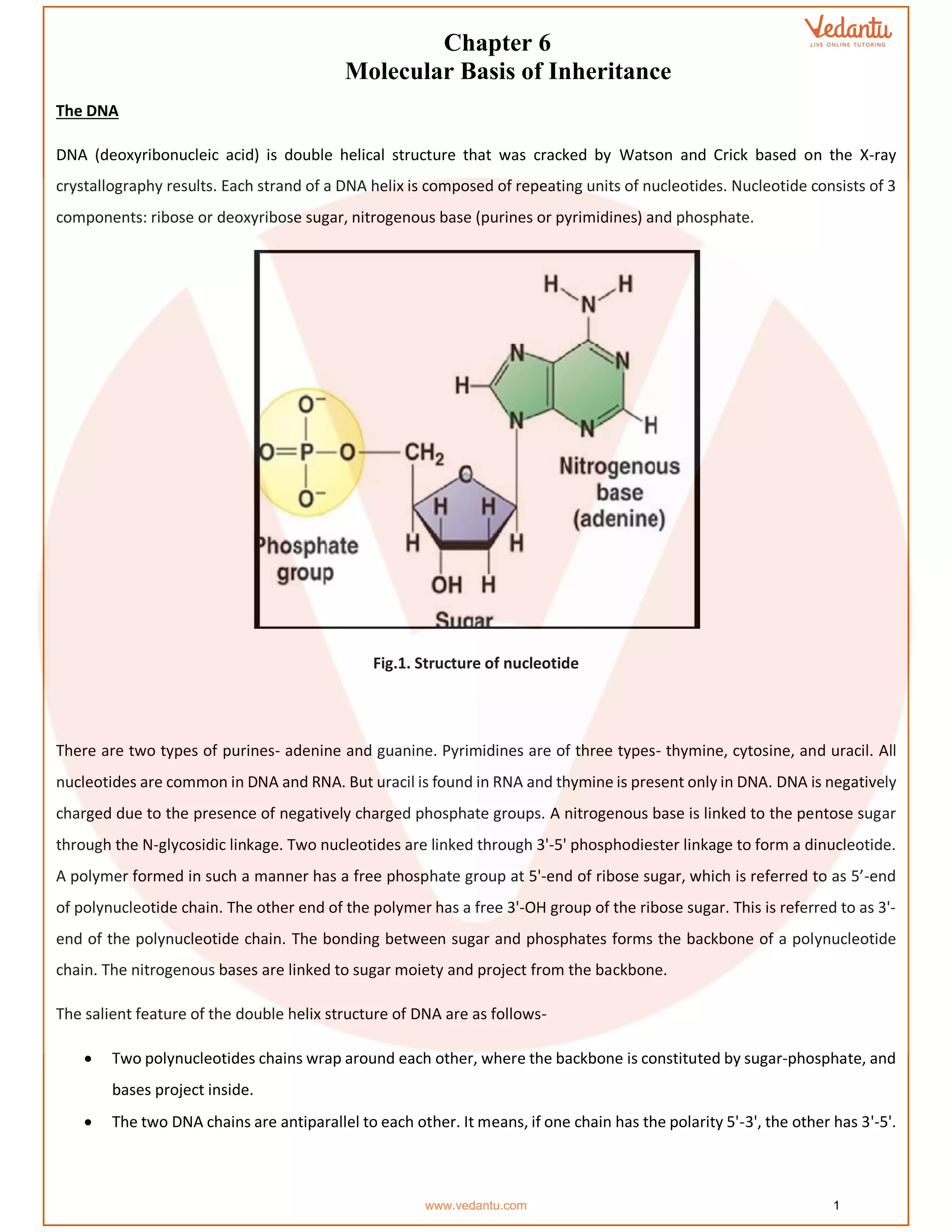
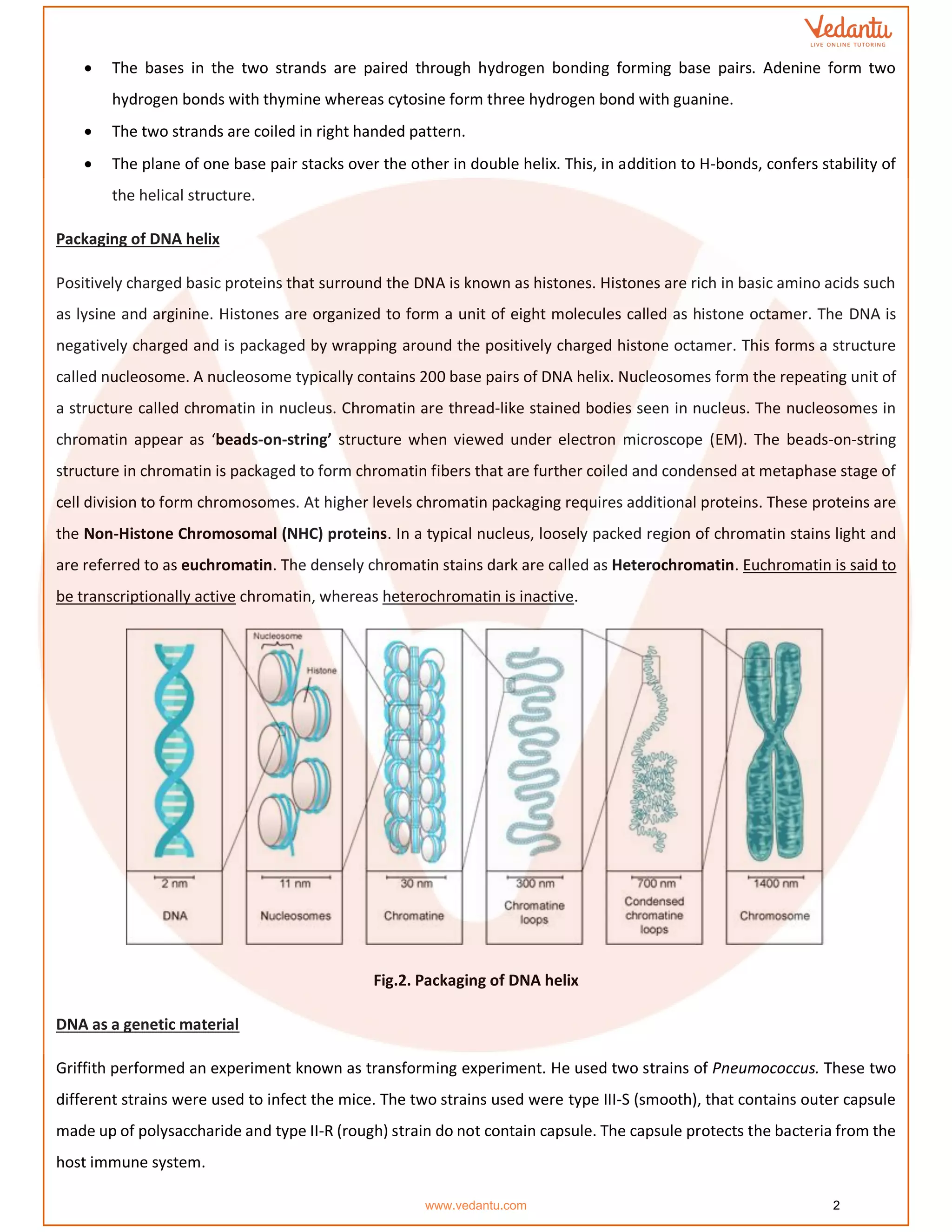
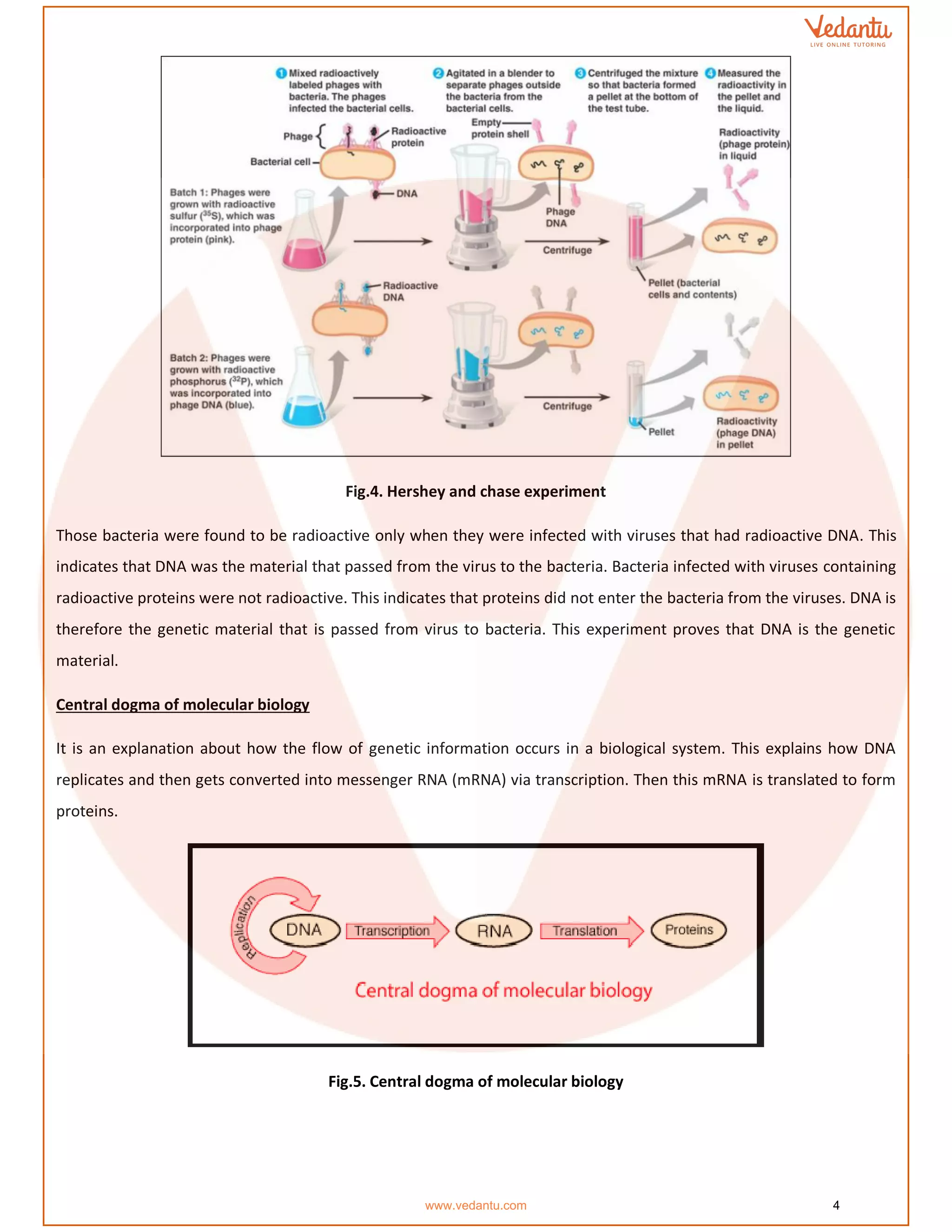
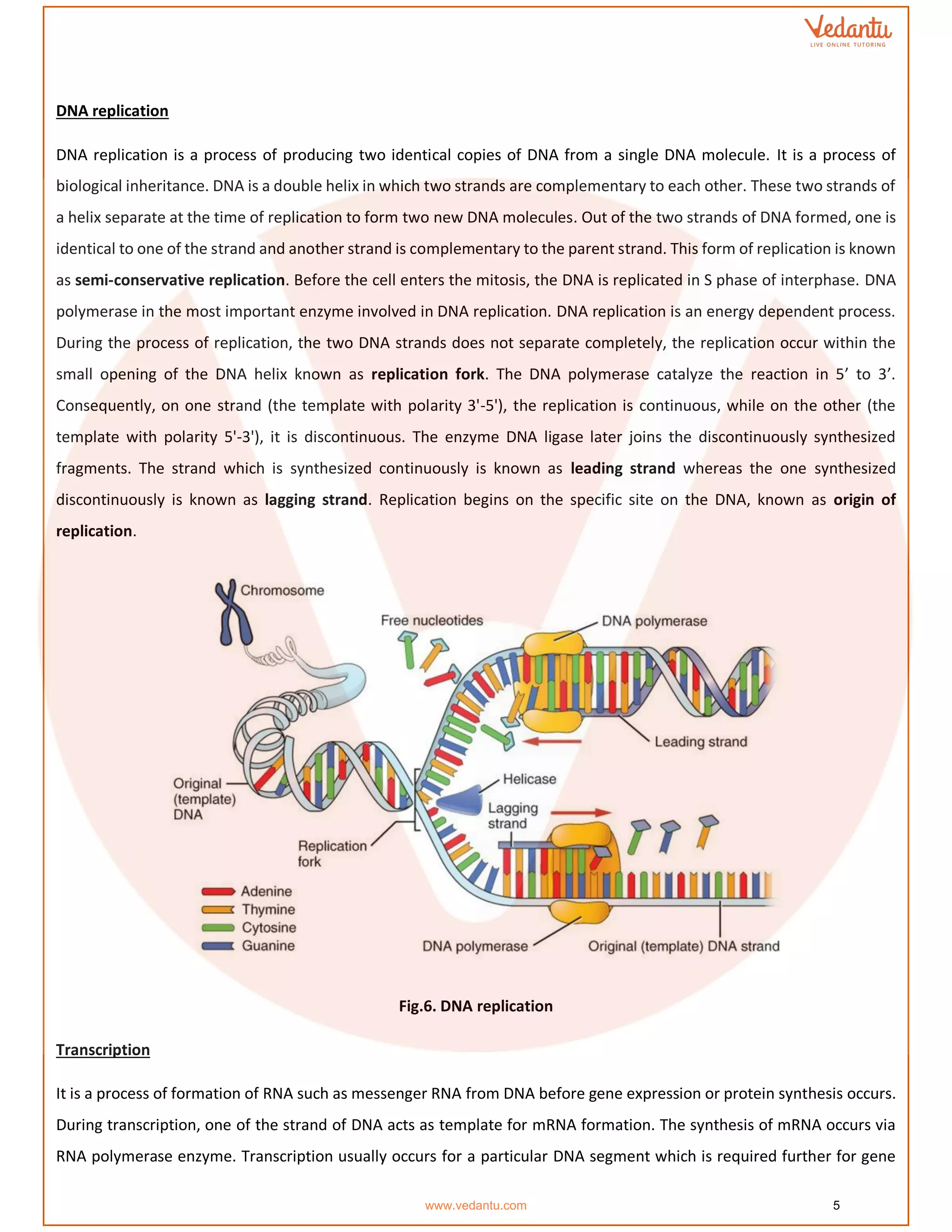
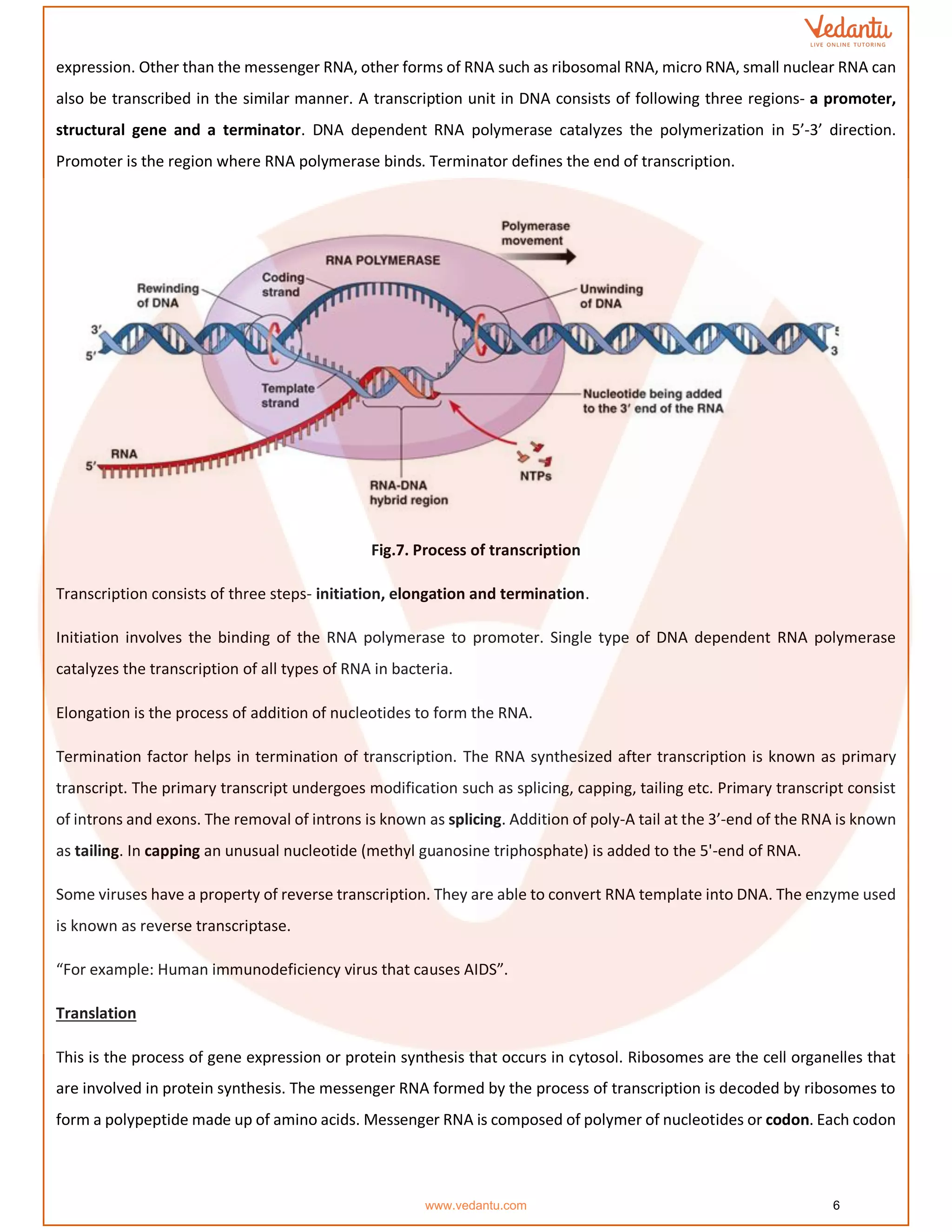
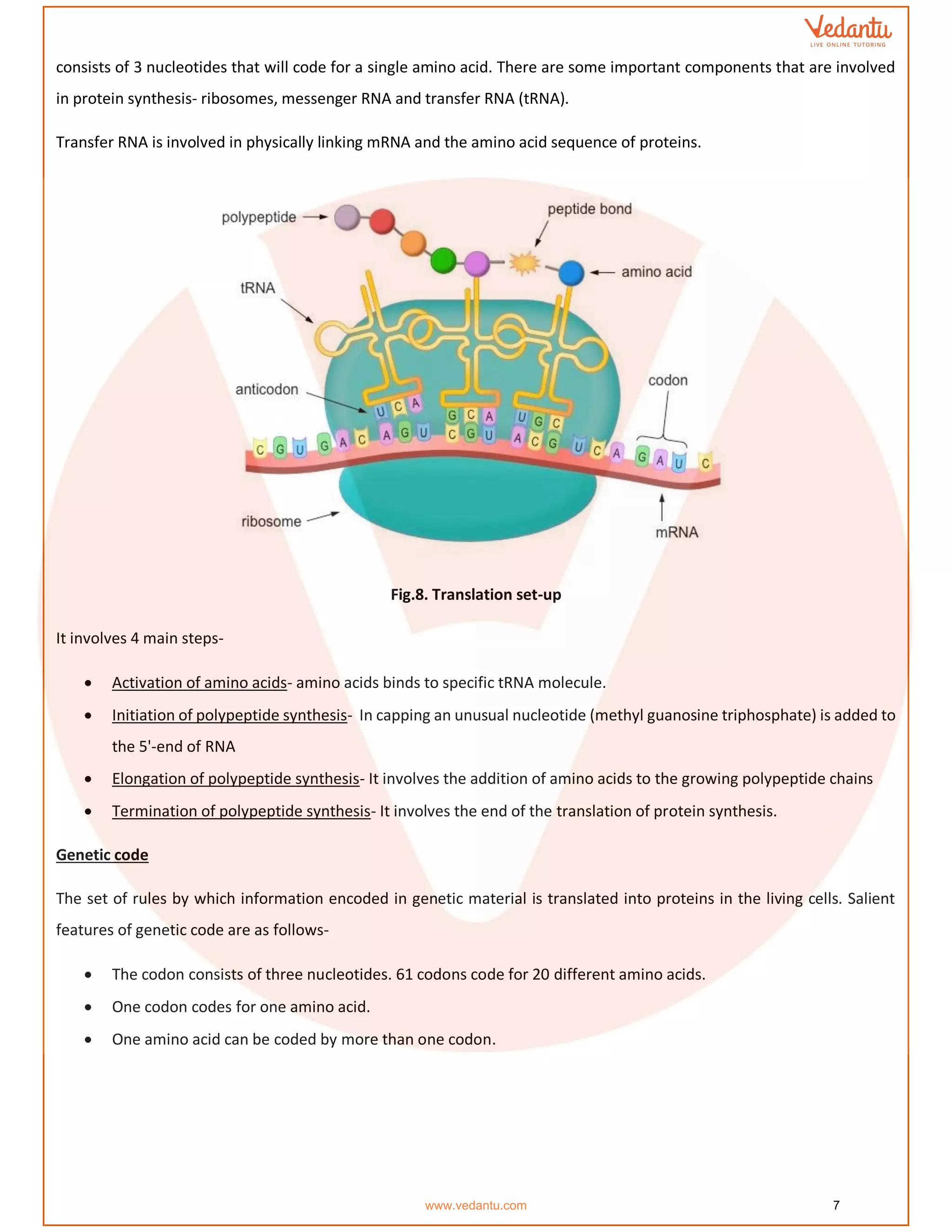
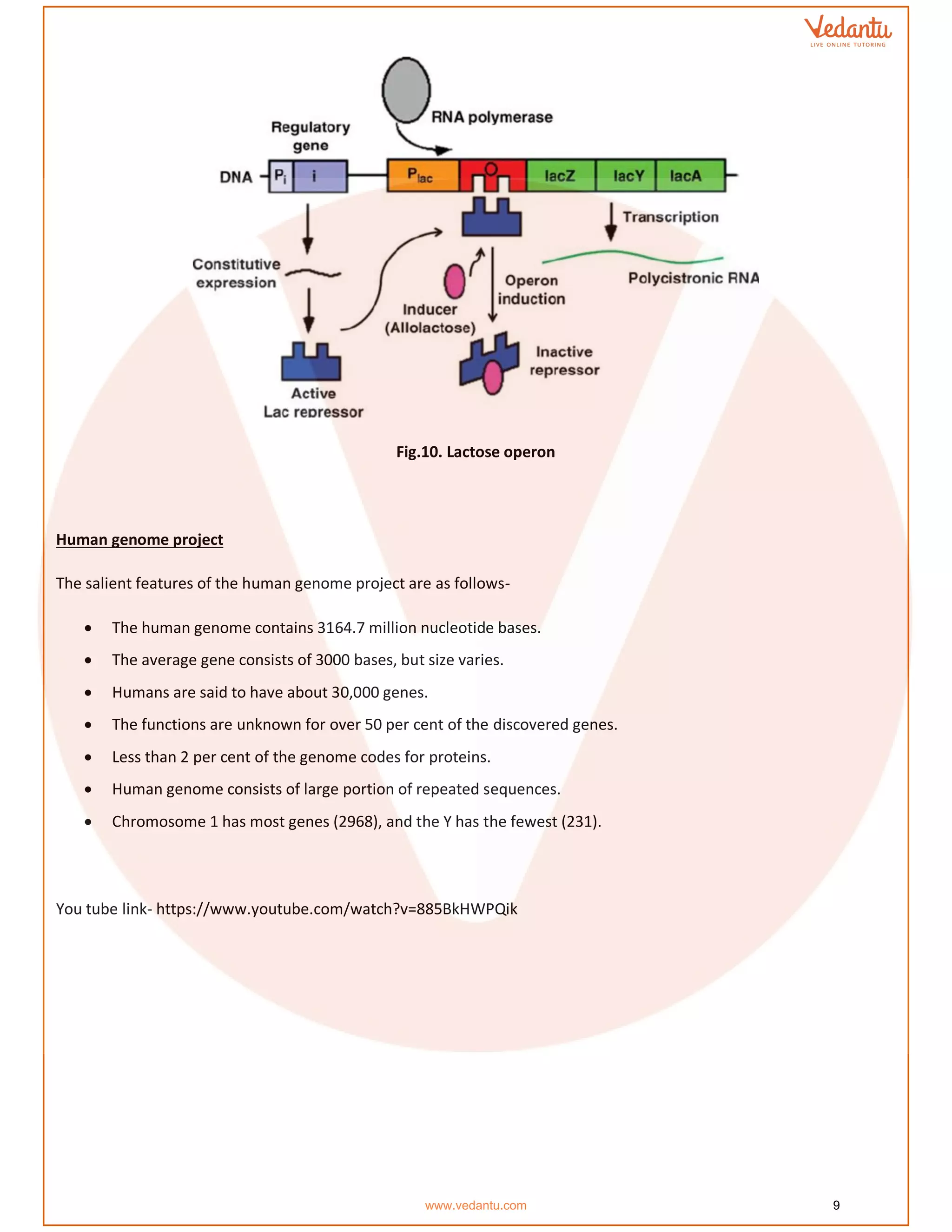
The document details the molecular basis of DNA, highlighting its structure, which consists of nucleotides made of sugar, phosphate, and nitrogenous bases. It discusses the packaging of DNA into chromatin and the experiments by Griffith and Hershey-Chase that demonstrate DNA as the genetic material. Additionally, it covers the central dogma of molecular biology, including DNA replication, transcription, and translation, along with the regulation of gene expression and a summary of the Human Genome Project.