Embed presentation



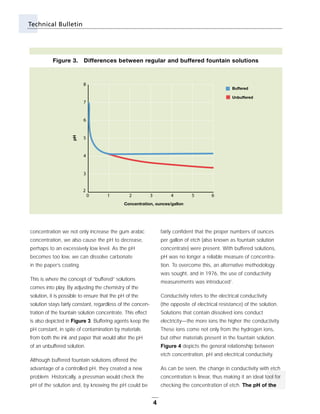
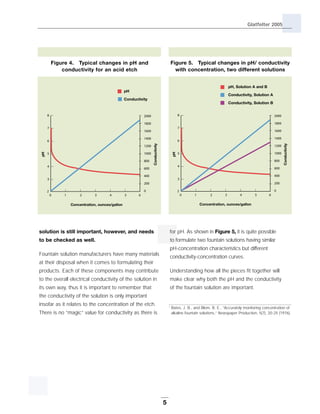



This technical bulletin discusses fountain solutions used in offset printing. It provides background on fountain solutions and their typical components, including acids, desensitizers, buffers, inhibitors and wetting agents. The document explains how pH and conductivity relate to fountain solution concentration and composition. It addresses how buffered solutions help maintain consistent pH levels and the importance of measuring both pH and conductivity. Frequently asked questions about alcohol use, conductivity guidelines, pH effects, and non-acidic fountain solutions are also covered.







