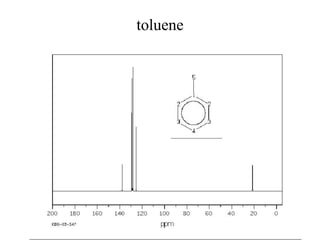
This document discusses carbon-13 nuclear magnetic resonance (13C NMR) spectroscopy. It begins by explaining the general theory of NMR, how nuclei absorb and emit radio waves when placed in a magnetic field. It then focuses on 13C NMR, noting that the number of signals correlates with the number of different carbon types in a molecule. Signal position depends on electron shielding, and chemical shift is expressed in parts per million. The document provides examples of 13C NMR spectra of various organic compounds and explains how distortionless enhancement by polarization (DEPT) distinguishes carbon types.