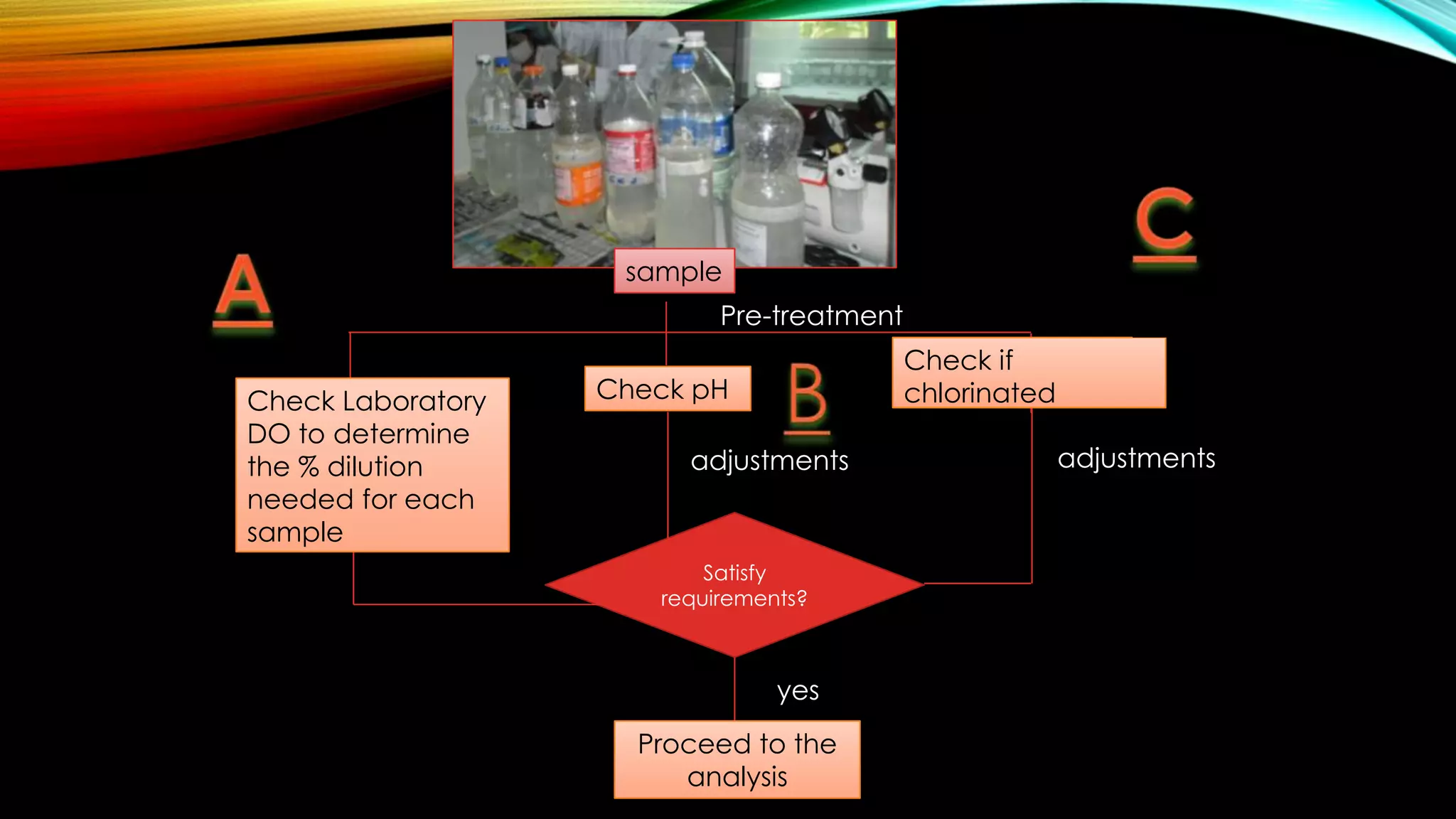
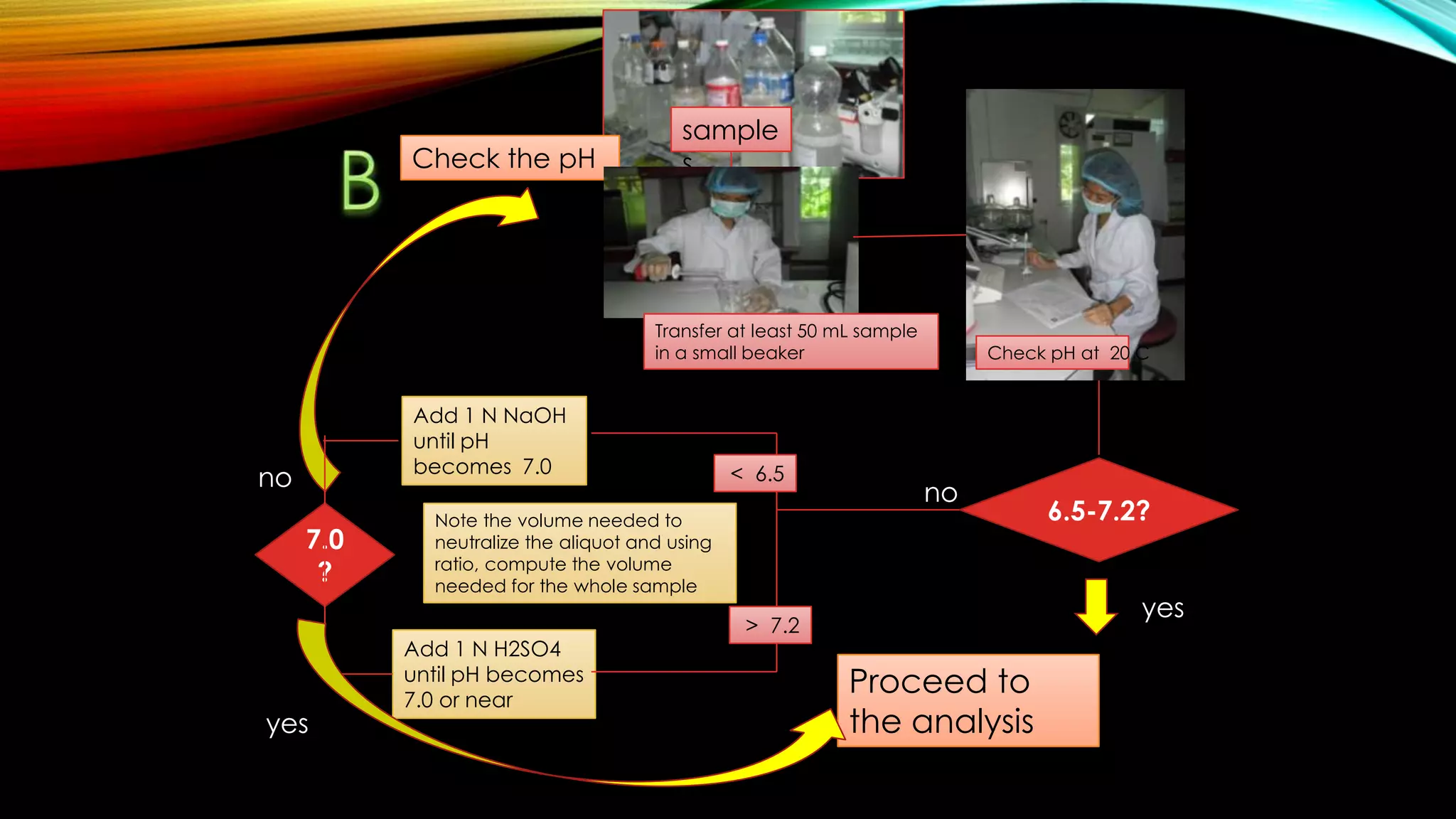
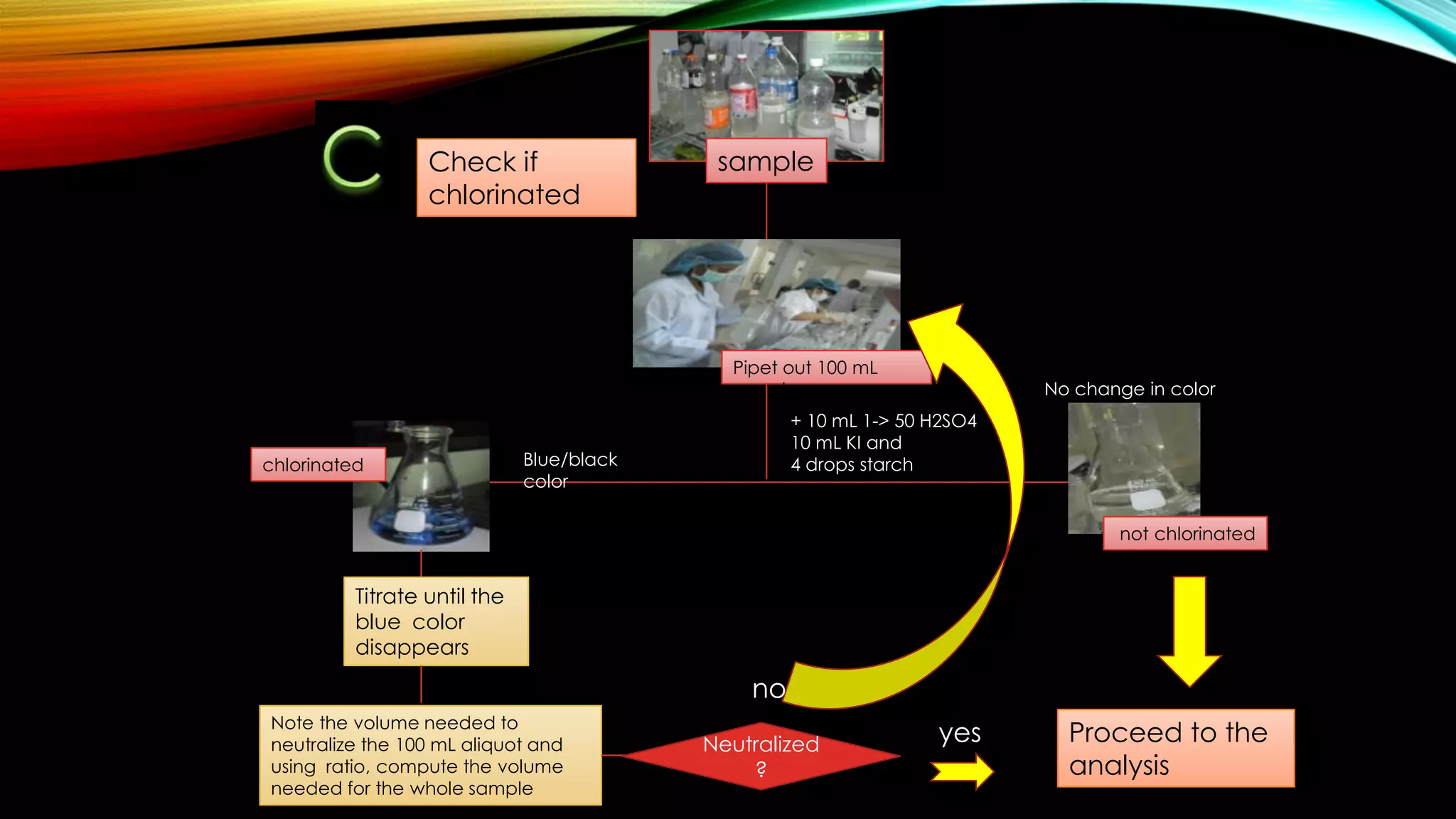
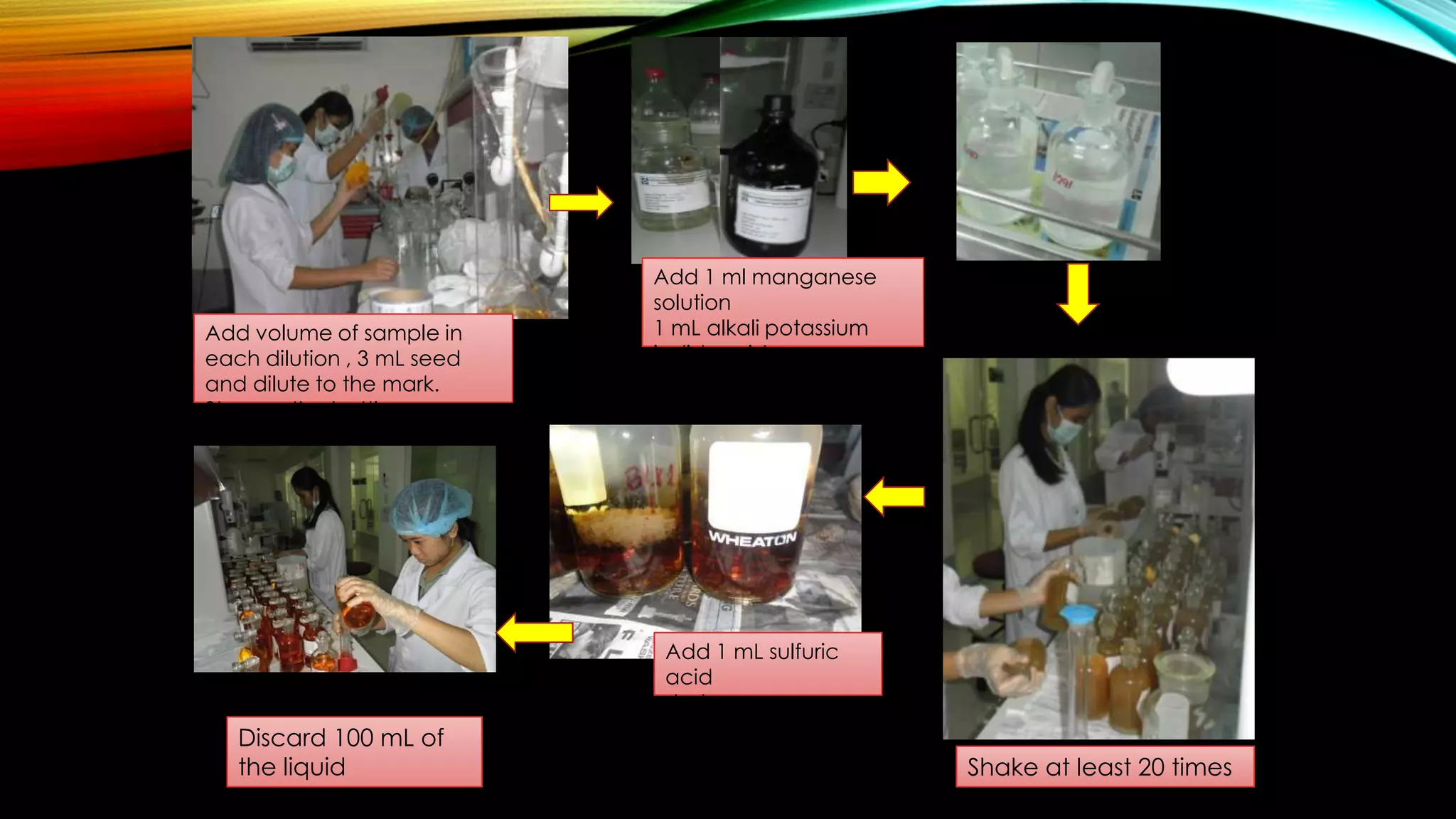
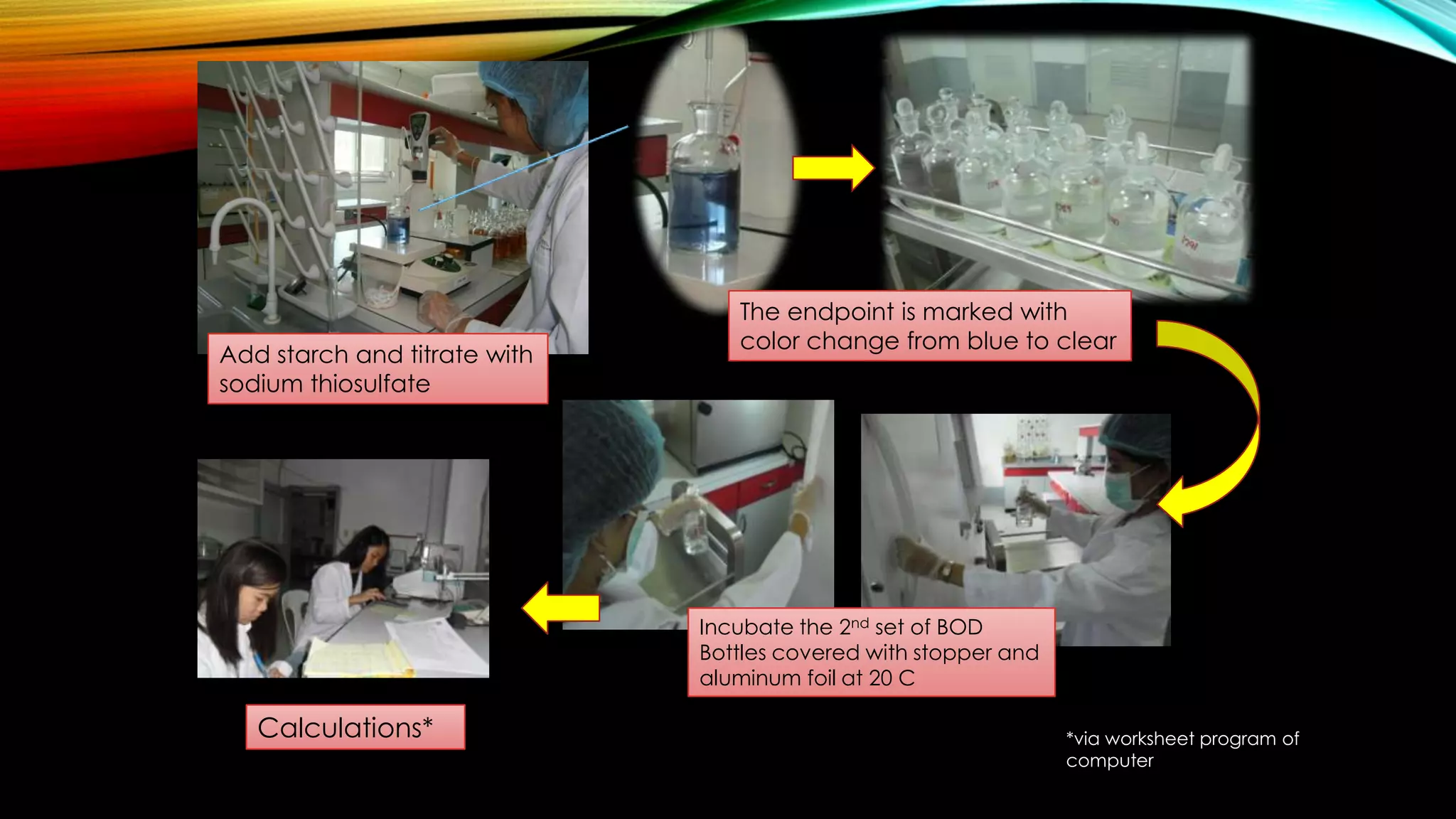
The document outlines the procedure for conducting a Biochemical Oxygen Demand (BOD) analysis, including pre-treatment steps to ensure sample suitability. It details laboratory requirements, dilution calculations, and titration methods to assess whether the water samples are chlorinated or not. The analysis requires careful monitoring of pH levels and involves specific treatments to prepare samples for accurate oxygen demand measurement.