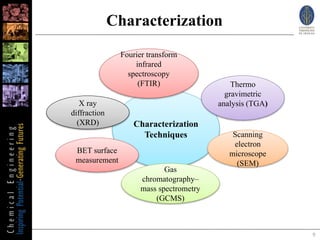
This document summarizes research on synthesizing solketal from glycerol and acetone using heterogeneous clay catalysts. Key points:
1) Aluminum pillared clay and montmorillonite clay catalysts were used to catalyze the reaction of glycerol and acetone to produce solketal.
2) Testing different reaction temperatures found that aluminum pillared clay performed best at 25°C, yielding 60% solketal, while montmorillonite yielded 58% at 40°C.
3) Characterization of the product confirmed the structure of solketal matched the commercial standard, demonstrating the synthesized solketal is effective.











![• Band 3432 cm−1 reveals axial
deformation in (-O-H) bond.
• 2986 and 2983 cm−1 refers to axial
deformation of methyl bonds (-C-
H) .
• 1456 cm−1 band showed water
angular deformation.
• band at 1372 cm−1 belongs to
ketone group methyls.
• Band 1211 cm−1 and 1046 cm−1
represents the dioxolane (five-
membered ring) -C-O bonds.
• bands 1158 cm−1 represents
solketal bonds (-C-O-C-O-C-) [6].
13
Results and Discussions
Fig 6.FTIR graph of sample product
compare with Sigma Aldrich Sample.](https://image.slidesharecdn.com/imtisalicost2020paperid147-220915073718-427edbe4/85/Biofuel-additives-ppt-13-320.jpg)


