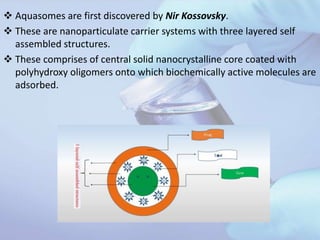
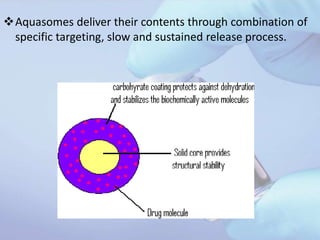
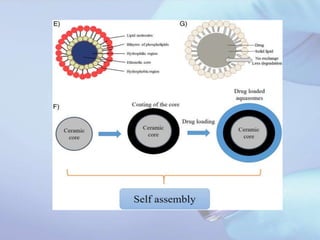
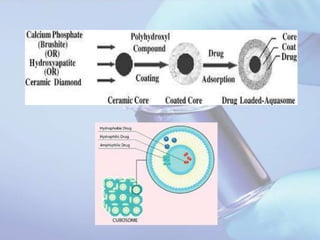
Aquasomes are innovative nanoparticulate carrier systems with a three-layer structure designed to deliver bioactive molecules while preserving their integrity. They are made by encapsulating a solid nanocrystalline core with polyhydroxy oligomers, allowing for targeted and sustained release of therapeutic agents, such as proteins and peptides. The preparation involves a three-step self-assembly process: core formation, coating, and drug immobilization, with various characterization techniques employed to analyze their properties.