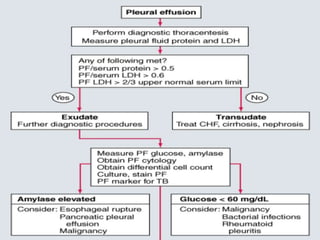
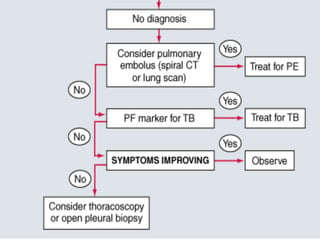
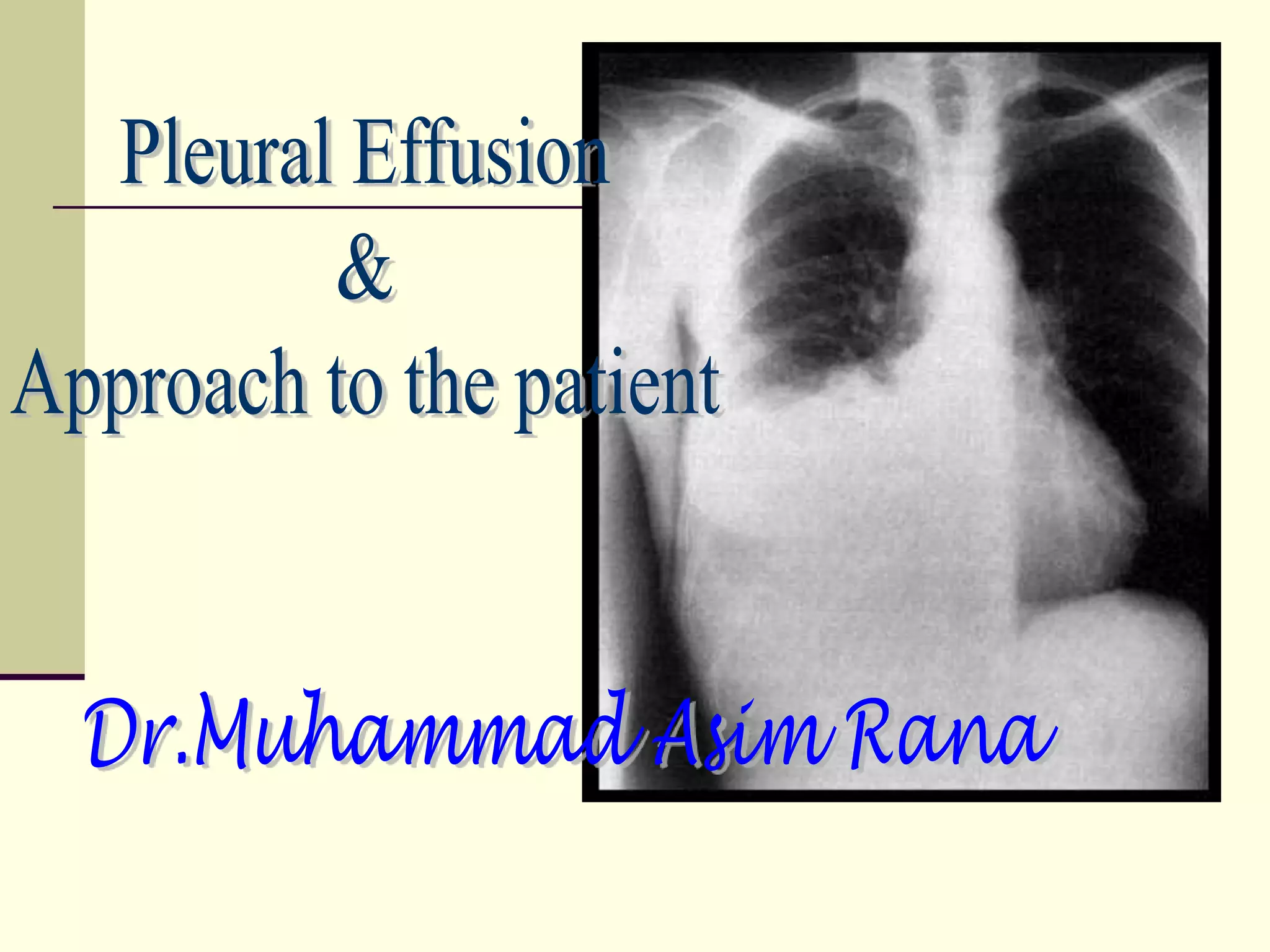
Pleural effusion occurs when excess fluid accumulates in the pleural space between the lungs and chest wall. A pleural effusion is considered an exudate if fluid formation exceeds lymphatic absorption, usually due to local inflammation, infection, or malignancy. Diagnostic evaluation involves determining if the effusion is a transudate or exudate based on pleural fluid analysis and comparing values to serum. Additional tests on exudative fluid including cell count, cultures, pH, and cytology aim to identify the specific cause. Treatment focuses on resolving the underlying condition causing fluid accumulation or drainage in some exudative cases.




![Transudative pleural effusions
result from alteration of hydrostatic and
oncotic factors that increase the formation or
decrease the absorption of pleural fluid (e.g.,
increased mean capillary pressure [heart
failure] or decreased oncotic pressure
[cirrhosis or nephrotic syndrome]).](https://image.slidesharecdn.com/approachtopleuraleffusion-140205151929-phpapp01-230921040041-080779fb/85/approachtopleuraleffusion-140205151929-phpapp01-pdf-6-320.jpg)