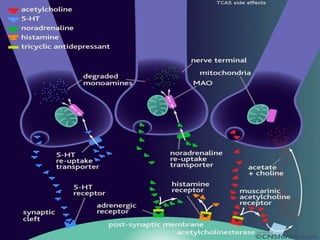
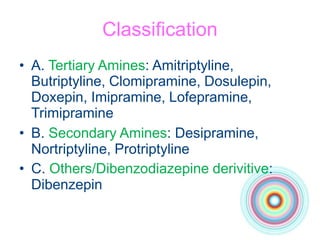
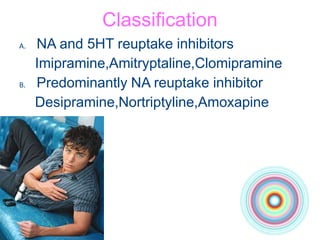
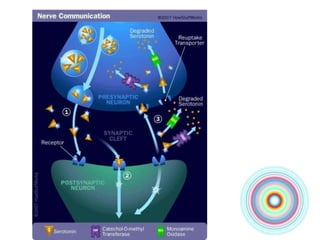
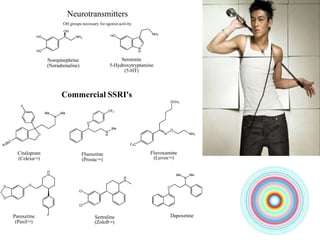
This document discusses antidepressants, including tricyclic antidepressants and selective serotonin reuptake inhibitors. It provides definitions of affective disorders and describes the neurobiological theory of depression. It discusses the pharmacology, indications, mechanisms of action, adverse effects and interactions of tricyclic antidepressants. It also discusses the pharmacology, mechanisms of action, important drugs, adverse effects and interactions of selective serotonin reuptake inhibitors. The document provides information on the treatment of depression and compares older tricyclic antidepressants to newer selective serotonin reuptake inhibitors.