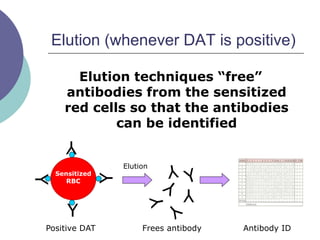
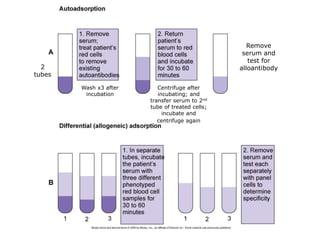
This document provides an overview of antibody identification in blood banking. It discusses:
1) The basics of antibody screens and the need to identify unexpected antibodies to ensure transfusion safety.
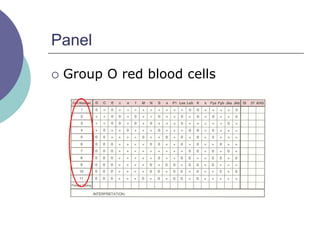
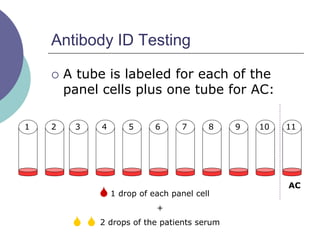
2) Key concepts like using patient serum as the unknown and reagent RBCs as knowns.
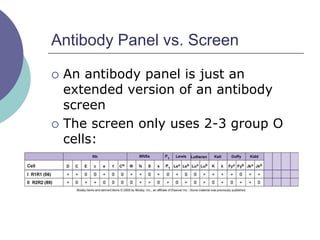
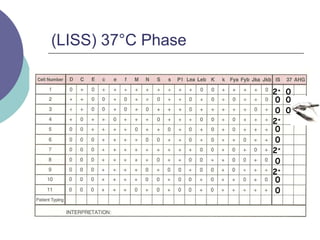
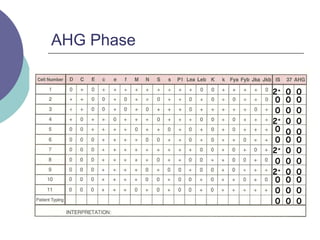
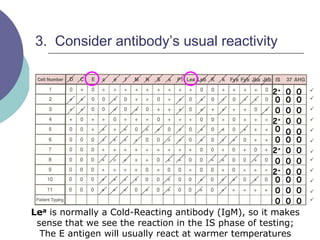
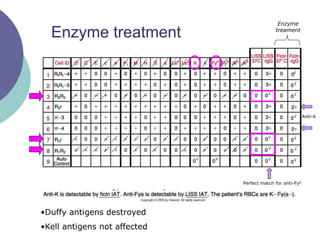
3) How antibody panels are performed using at least 10 RBCs and techniques like the immediate spin, 37C incubation, and antiglobulin phases.
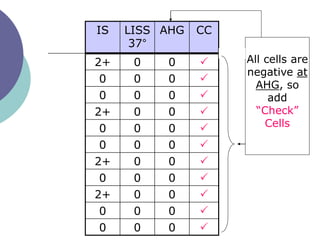
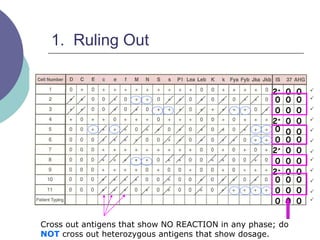
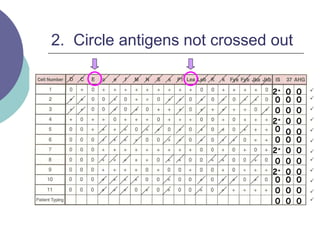
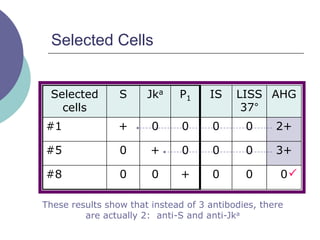
4) Guidelines for interpreting panels by ruling out non-reactive antigens, circling reactive ones, considering antibody characteristics, and looking for a matching pattern to identify the antibody.