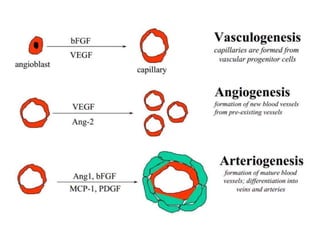
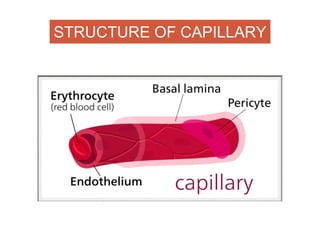
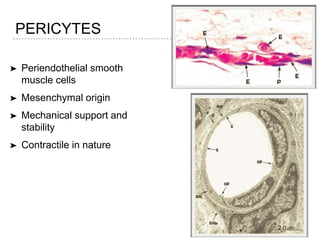
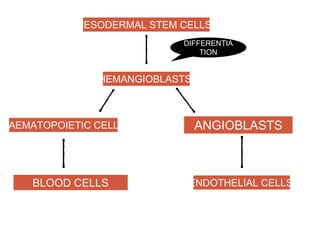
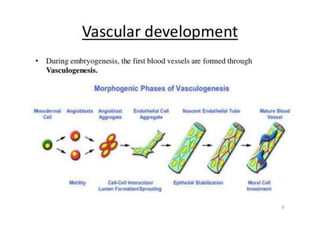
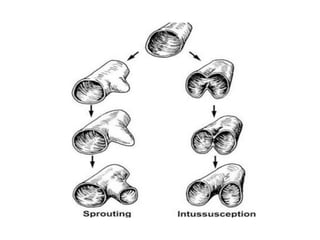
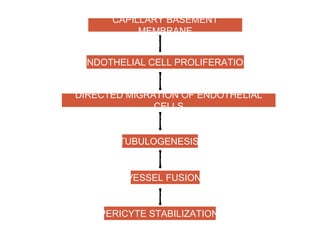
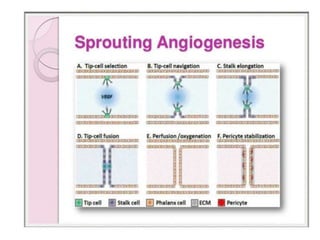
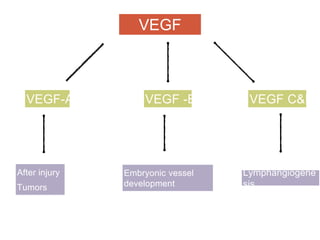
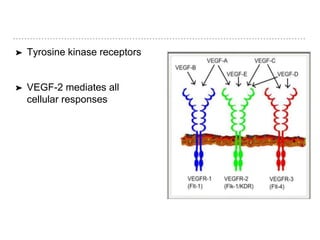
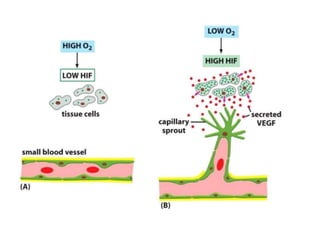
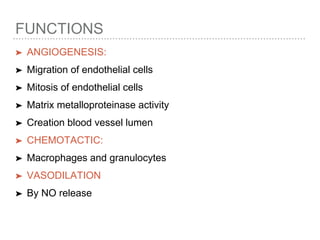
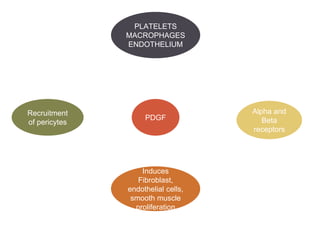
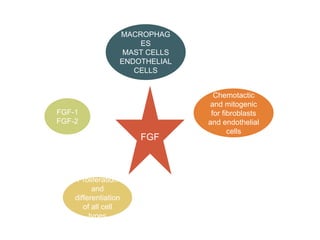
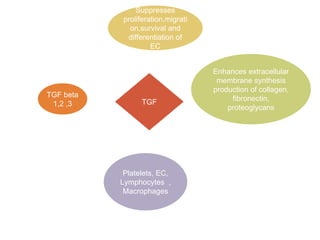
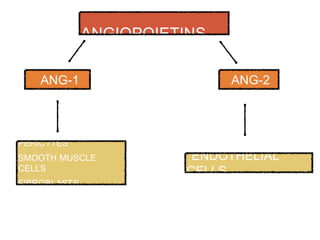
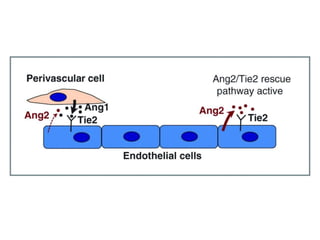
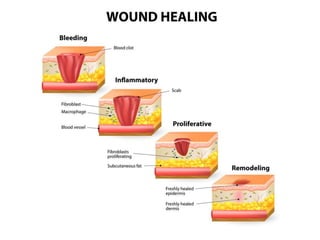
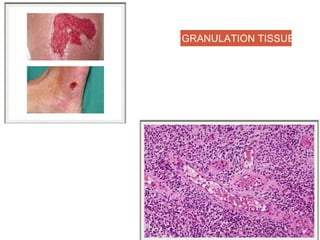
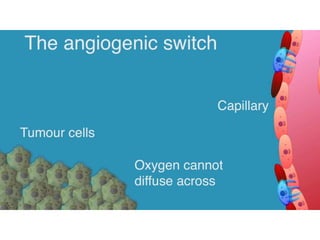
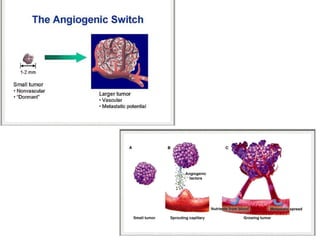
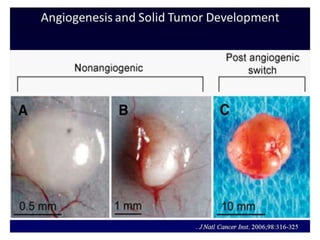
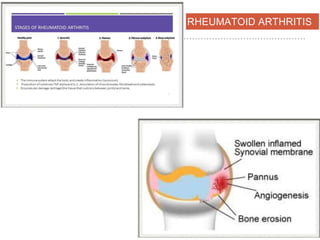
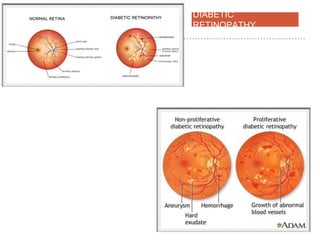
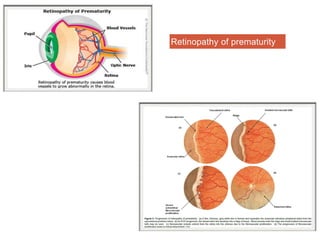
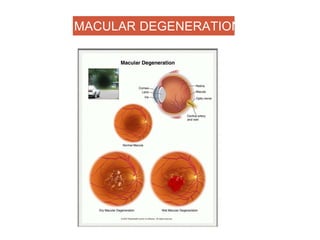
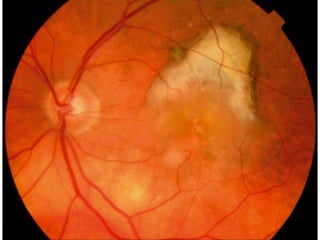
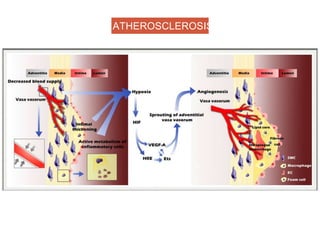
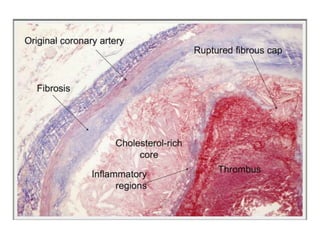
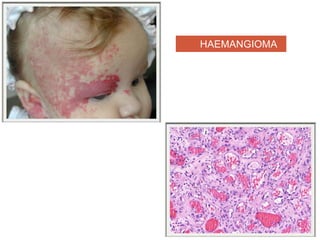
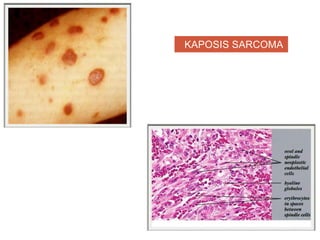
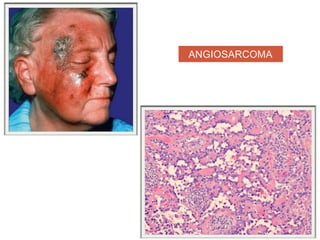
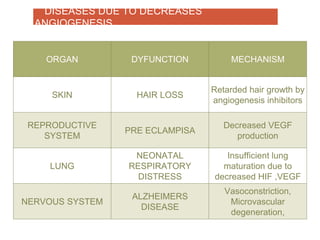
This document discusses angiogenesis, the process of forming new blood vessels from existing ones, highlighting its significance in both health and disease, including its role in wound healing and tumor growth. Key factors and mechanisms involved in angiogenesis, such as VEGF and different angiogenic processes, are detailed, along with the effects of angiogenic inhibitors and potential therapeutic applications. The text also explores the relationship between angiogenesis and various conditions, emphasizing its clinical relevance for future disease management.